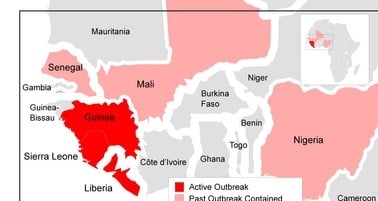
On Dec. 26, 2013, a two-year-old boy living in the Guinean village of Meliandou, Guéckédou Prefecture, was stricken with a rare disease, caused by the filament-shaped Ebola virus.
The child is believed to be the first case in what soon became a flood-tide of contagion, ravaging the West African countries of Guinea, Sierra Leone and Liberia, infecting over 21,000 cases as of Jan. 21, according to the World Health Organization, with nearly 9,000 confirmed deaths – the actual toll likely much higher.
Now, researchers from Arizona State University and Georgia State University are trying to better understand the epidemiology of the Ebola virus disease in order to alleviate suffering and prevent future disease outbreaks from reaching the catastrophic proportions of the current crisis.
In a report appearing in the February 2015 issue of the prestigious British medical journal The Lancet Infectious Diseases, ASU researchers present the study “Modelling the effect of early detection of Ebola,” which examines the levels of detection and patient isolation required to shut down transmission of Ebola and reports on new efforts to model the impact of timely diagnostic testing on the spread of Ebola across populations.
A better understanding of viral dissemination and techniques for disease management are vital if a similar calamity is to be avoided in the future.
Wave of destruction
The Ebola virus has become notorious, not only for its highly contagious and lethal nature, but for the nightmarish assortment of symptoms collectively known as hemorrhagic fever. These may include vomiting of blood, bleeding from the eyes, ears, nose, mouth, rectum, internal bleeding,excruciating pain and the liquidization of internal organs.
The three West African nations centrally affected by the epidemic were acutely unprepared for the crisis. Treatment centers were rapidly swamped with severely ill patients. Resources for proper care, isolation of infected patients and even basic means of sterilization were soon depleted. Health care workers were especially vulnerable to infection.
A number of exacerbating factors contributed to the outbreak and rapid spread of Ebola in the region. Timber and mining activities have impacted densely forested regions and brought fruit bats – believed to be a natural reservoir for the virus – in closer contact with humans. Infected animals consumed as bush meat may also have planted early seeds of the disease in the vulnerable population. Long periods of civil unrest have left the area deeply impoverished and the health infrastructure fractured.
Meliandou, the town identified as ground zero, is situated in a forested area at the convergence point of Guinea, Liberia and Sierra Leone. Populations move fluidly across these porous borders, as impoverished residents are often on the move in search of work. These conditions created a perfect storm for the aggressive virus.
An additional factor fueling the explosive spread of Ebola in West Africa was the delayed and inadequate response to the crisis on the part of developed countries and global health organizations.
Time is the enemy
As the authors of the current study emphasize, breaking the chain of Ebola transmission presents intimidating challenges. After the development of symptoms, the virus is highly contagious, and each new contact presents an opportunity for further spread of the disease.
Tracking all contacts of infected individuals can be a daunting effort, even in first-world settings, with low case numbers. In the absence of a vaccine or reliable therapeutic for Ebola, diagnosis of the disease at a pre-symptomatic stage and rapid isolation of infected individuals are the surest means for arresting further disease transmission.
“Early detection of Ebola infection provides the opportunity and time to safely isolate and treat individuals before they become contagious,” said Karen Anderson, Biodesign Institute researcher and professor in ASU’s School of Life Sciences. “Our findings show two key things: first, that the predicted impact of early diagnostic tests depends on existing public health measures. Second, there appears to be a tipping point, where early diagnosis of high-risk individuals, combined with adequate isolation, can markedly decrease the predicted number of infected individuals.”
Stopping an epidemic in its tracks requires a reduction in a critical value known as the reproductive ratio – a measure of new infections generated by a single case over the course of the infectious period. The higher the number for the reproductive ratio, the more difficult an epidemic is to contain.
A technique known as polymerase chain reaction (PCR) can be used for pre-symptomatic identification of the Ebola virus. The current study models the expected outcomes on viral transmission of Ebola using PCR-based pre-symptomatic diagnosis and isolation of infected patients within three days of the onset of symptoms.
“Our results underscore the dramatic impact that diagnostic capacity can bring about during an Ebola epidemic to quickly identify Ebola cases before these start new chains of transmission in the community or health care settings,” said Diego Chowell, lead author of the study.
Carlos Castillo-Chavez, director of the Simon A. Levin Mathematical, Computational and Modeling Sciences Center, emphasizes the power of mathematical modeling for understanding and limiting the scale of epidemics: “Finding that small differences in isolation effectiveness may have a large impact on epidemic size highlights the importance of evaluating novel diagnostic technologies at the population level using mathematical models,” he says. “An intervention may not work or be effective unless it is effectively used beyond a tipping point.”
The authors urge the implementation of the strategy of pre-symptomatic diagnosis and rapid isolation, targeting high-risk individuals, including care givers and health care workers.
The desperate need for early diagnosis of Ebola is clear. During the West African epidemic, most Ebola patients remained undiagnosed in their communities. Among those who were diagnosed, the average time from symptom onset to positive diagnosis was about five days – a prescription for rapid, far-flung transmission of the disease.
End game
According to the latest reports from the World Health Organization, the Ebola epidemic appears to be weakening its grip on the region. For the first time since June 2014, there have been fewer than 100 new weekly cases reported in the three countries most affected, signaling what health care workers hope is the final phase of Ebola’s devastating reign.
Increased vigilance and new tools at both the epidemiological and therapeutic ends of the spectrum are vitally needed if another epidemic – perhaps of even greater scale – is to be prevented.