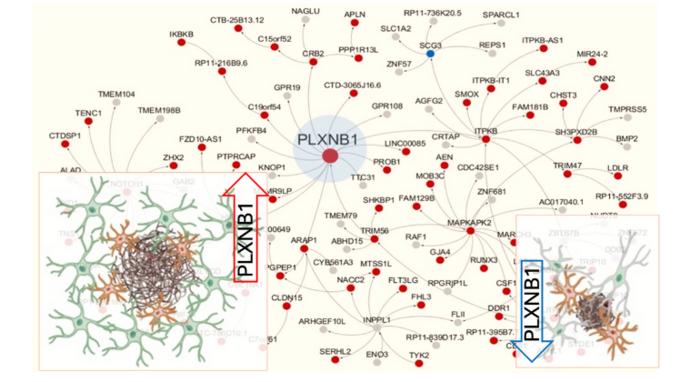
Researchers have found a new way to potentially slow or stop Alzheimer’s disease. Their study shows that the plexin-B1 protein and reactive astrocytes (a type of brain cell) are key in managing amyloid plaques, which are linked to Alzheimer’s. By manipulating plexin-B1, they found that brain cells can better clear these plaques. The research provides new insights into brain cell communication and opens possibilities for innovative treatments.
Researchers at the Icahn School of Medicine at Mount Sinai say they have made a significant breakthrough in Alzheimer’s disease research by identifying a novel way to potentially slow down or even halt disease progression.
The study, which focuses on the role of reactive astrocytes and the plexin-B1 protein in Alzheimer’s pathophysiology, provides crucial insights into brain cell communication and opens the door to innovative treatment strategies. It was published in Nature Neuroscience on May 27.
This groundbreaking work is centered on the manipulation of the plexin-B1 protein to enhance the brain’s ability to clear amyloid plaques, a hallmark of Alzheimer’s disease. Reactive astrocytes, a type of brain cell that becomes activated in response to injury or disease, were found to play a crucial role in this process. They help control the spacing around amyloid plaques, affecting how other brain cells can access and clear these harmful deposits. “Our findings offer a promising path for developing new treatments by improving how cells interact with these harmful plaques,” said Roland Friedel, PhD, Associate Professor of Neuroscience, and Neurosurgery, at Icahn Mount Sinai and a senior author of the study. The research was driven by the analysis of complex data comparing healthy individuals to those with Alzheimer’s, aiming to understand the disease’s molecular and cellular foundations.
Hongyan Zou, PhD, Professor of Neurosurgery, and Neuroscience, at Icahn Mount Sinai and one of the study’s lead authors, highlighted the broader implications of their findings: “Our study opens new pathways for Alzheimer’s research, emphasizing the importance of cellular interactions in developing neurodegenerative disease treatments.”
One of the study’s most significant achievements is its validation of multiscale gene network models of Alzheimer’s disease. “This study not only confirms one of the most important predictions from our gene network models but also significantly advances our understanding of Alzheimer’s. It lays a solid foundation for developing novel therapeutics targeting such highly predictive network models,” said Bin Zhang, PhD, Willard T.C. Johnson Research Professor of Neurogenetics at Icahn Mount Sinai and one of the study’s lead authors. By demonstrating the critical role of plexin-B1 in Alzheimer’s disease, the research underscores the potential of targeted therapies to disrupt the disease’s progression.
The research team emphasizes that while their findings mark a significant advance in the fight against Alzheimer’s, more research is needed to translate these discoveries into treatments for human patients.
“Our ultimate goal is to develop treatments that can prevent or slow down Alzheimer’s progression,” Dr. Zhang added, outlining the team’s commitment to further exploring the therapeutic potential of plexin-B1.
This study is supported by the NIH National Institute on Aging (NIA) grants U01AG046170 and RF1AG057440 and is part of the NIA-led Accelerating Medicines Partnership – Alzheimer’s Disease (AMP-AD) Target Discovery and Preclinical Validation program. This public private partnership aims to shorten the time between the discovery of potential drug targets and the development of new drugs for Alzheimer’s disease treatment and prevention.
The paper is titled “Regulation of cell distancing in peri-plaque glial nets by Plexin-B1 affects glial activation and amyloid compaction in Alzheimer’s disease.”