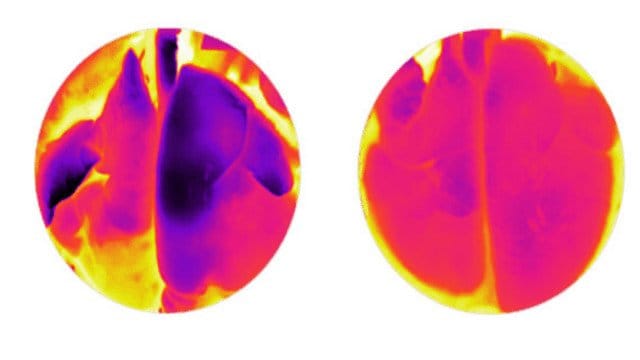
Lung transplantation, the only life-saving therapy for an increasing population of patients with end-stage lung disease, is severely limited by the number of available donor organs. Currently, up to 80% of donor lungs are rejected for serious but potentially reversible injuries. Since the beginning of transplantation in 1960s, clinicians and scientists have been trying to address the critical shortage of donor organs.
Now, a multidisciplinary team from Columbia Engineering and Vanderbilt University has—for the first time—demonstrated in a clinically relevant model that severely damaged lungs can be regenerated to meet transplantation criteria. In a study published today on Nature Communications’ website, the researchers describe the cross-circulation platform that maintained the viability and function of the donor lung and the stability of the recipient for 36 to 56 hours. As Brandon Guenthart, a lead author of the study, explains “to support lung recovery and to demonstrate cellular regeneration, we had to pursue a radically different approach and develop more minimally invasive diagnostics.” Current methodologies of lung support are limited to only 6 to 8 hours, a time that is too short for therapeutic interventions that could regenerate the injured lung and improve its function.
The team, co-led by Gordana Vunjak-Novakovic, University Professor and The Mikati Foundation Professor of Biomedical Engineering and Medical Sciences at Columbia Engineering, and Matthew Bacchetta, the H. William Scott Professor of Surgery at Vanderbilt University and adjunct professor at Columbia’s department of biomedical engineering, also developed new diagnostic tools for the non-invasive evaluation of the regenerating lung. They expect their advance will lead to an increase in the number of lungs for transplant, through the recovery of severely damaged lungs that are currently unsuitable for clinical use.
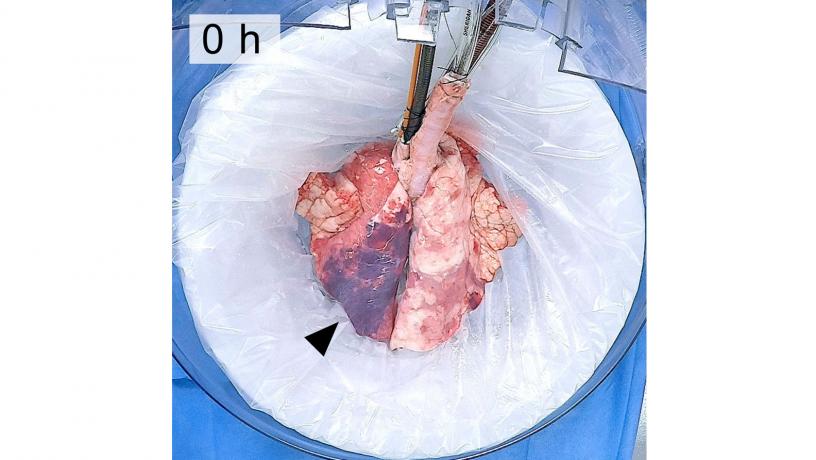
Macroscopic appearance of lungs throughout 36 hours of ex vivo support.
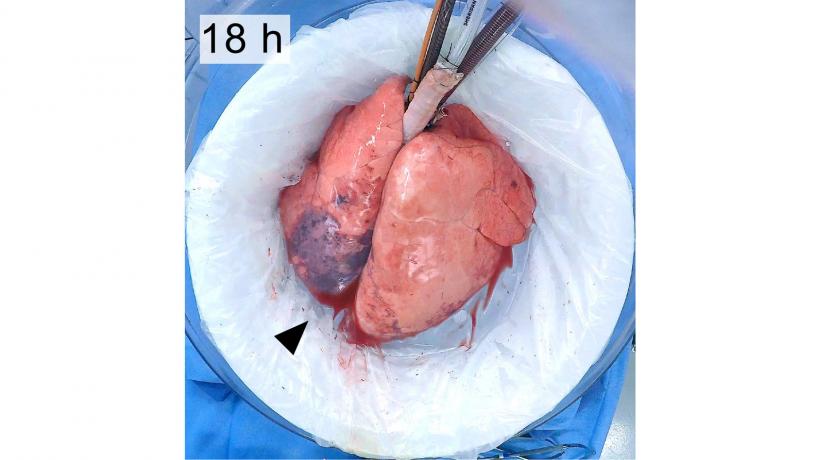
Macroscopic appearance of lungs throughout 36 hours of ex vivo support.
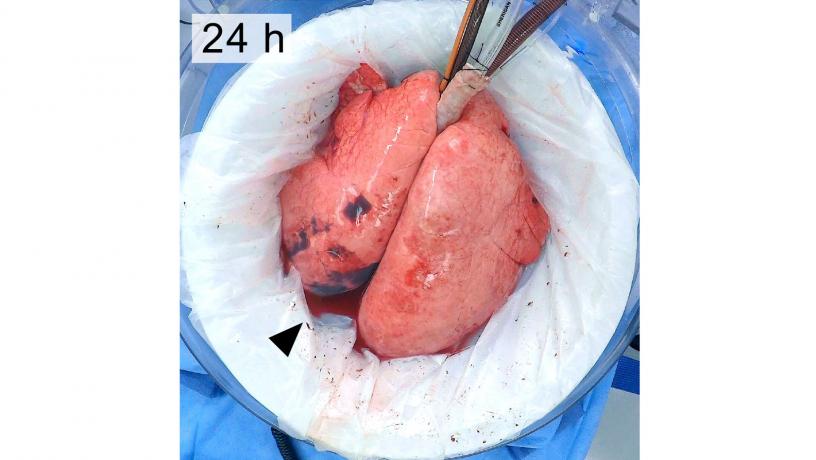
Macroscopic appearance of lungs throughout 36 hours of ex vivo support.
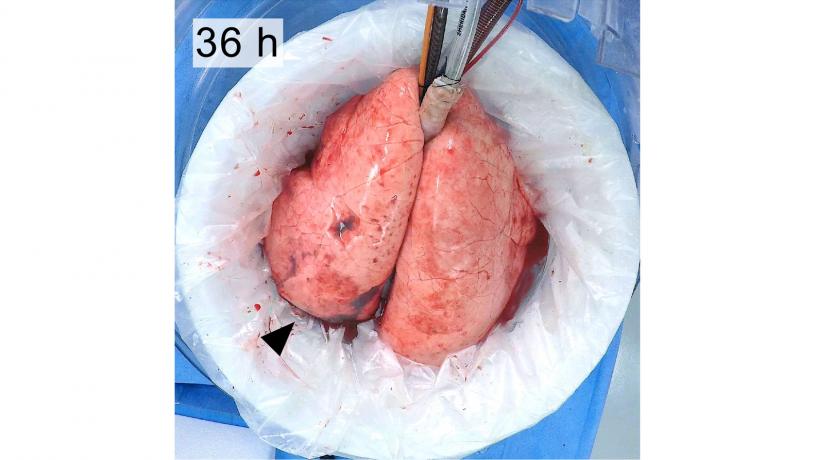
Macroscopic appearance of lungs throughout 36 hours of ex vivo support.
A previous study from the team demonstrated a cross-circulation platform that maintained the viability and function of a donor lung for 36 hours. The researchers were able to use their advanced support system to fully recover the functionality of lungs injured by ischemia (restricted blood supply) and make them suitable for transplant.
For this new study, the team decided to test the effectiveness of their platform technology combined with conventional therapies and new diagnostics on lungs afflicted by the most frequent injury leading to donor lung rejection—gastric aspiration. This injury is caused by the entry of gastric material into the respiratory tract, resulting in severe injury to the pulmonary epithelium and thus making the lung unacceptable for transplantation. Currently, severely damaged donor lungs cannot be salvaged using existing devices or methods. This new study suggests that lungs injured by gastric aspiration can be maintained outside the body for several days, are amenable to repeated therapeutic interventions, and display evidence of cellular regeneration and improved function. Lungs regenerated on this platform met all criteria for transplantation.
“For seven years, we have diligently worked to develop new technologies for the maintenance and recovery of donor organs. This paper represents a culmination of fundamental and translational studies of lung bioengineering that have converged into a system capable to recover severely damaged lungs. We now have the team and technology to bring this research to the patients, by making more donor lungs available for transplant,” says Vunjak-Novakovic.
The team plans to conduct further studies to evaluate the functional capacity of the lungs following transplantation and the safety of the method, using a clinically relevant large animal model with immunosuppression.
“We envision that interventional cross-circulation may be used to investigate regeneration of other damaged organs, such as hearts, kidneys, and livers, expanding donor pools by salvaging severely damaged organs and leading to more organ transplants,” Bacchetta adds.