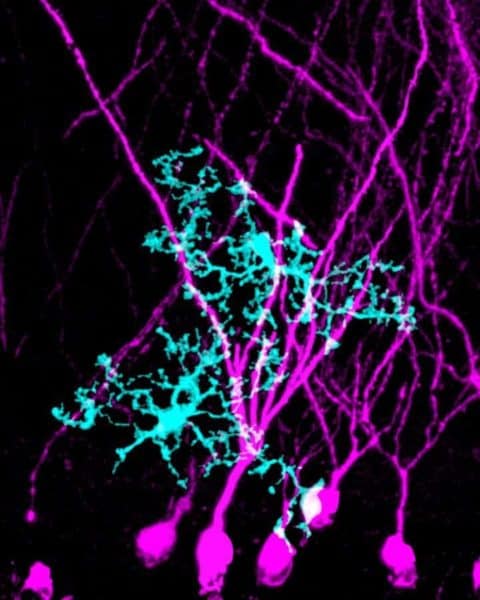
To make new memories, our brain cells first must find one another. Small protrusions that bud out from the ends of neurons’ long, branching tentacles dock neurons together so they can talk. These ports of cellular chatter – called synapses, and found in the trillions throughout the brain – allow us to represent new knowledge. But scientists are still learning just how these connections form in response to new experiences and information. Now, a study by scientists in the UCSF Weill Institute for Neurosciences has identified a surprising new way that the brain’s immune cells help out.
In recent years, scientists have discovered that the brain’s dedicated immune cells, called microglia, can help get rid of unnecessary connections between neurons, perhaps by engulfing synapses and breaking them down. But the new study, published July 1, 2020, in Cell, finds microglia can also do the opposite – making way for new synapses to form by chomping away at the dense web of proteins between cells, clearing a space so neurons can find one another. Continuing to study this new role for microglia might eventually lead to new therapeutic targets in certain memory disorders, the researchers say.
Neurons live within a gelatinous mesh of proteins and other molecules that help to maintain the three-dimensional structure of the brain. This scaffolding, collectively called the extracellular matrix (ECM), has long been an afterthought in neuroscience. For decades, researchers focused on neurons, and, more recently, the cells that support them, have largely considered the ECM unimportant.
But neurobiologists are starting to realize that the ECM, which makes up about 20 percent of the brain, actually plays a role in important processes like learning and memory. At a certain point in brain development, for example, the solidifying ECM seems to put the brakes on the rapid pace at which new neuronal connections turn over in babies, seemingly shifting the brain’s priority from the breakneck adaptation to the new world around it, to a more stable maintenance of knowledge over time. Scientists also wonder if a stiffening of the extracellular matrix later in life might somehow correspond to the memory challenges that come with aging.
“The extracellular matrix has been here the whole time,” said the study’s first author Phi Nguyen, a biomedical sciences graduate student at UCSF. “But it’s definitely been understudied.”
Nguyen and his adviser, Anna Molofsky, MD, PhD, an associate professor in the UCSF Department of Psychiatry and Behavioral Sciences, first realized the ECM was important to their research on the hippocampus, a brain structure critical for learning and memory, when an experiment yielded unexpected results. Knowing that microglia chew away at obsolete synapses, they expected that disrupting microglia function would cause the number of synapses in the hippocampus to shoot up. Instead, synapse numbers dropped. And where they thought they’d find pieces of synapses being broken down in the “bellies” of microglia, instead they found pieces of the ECM.
“In this case microglia were eating something different than we expected,” Molofsky said. “They’re eating the space around synapses – removing obstructions to help new synapses to form.”
Before springing into action, the microglia wait for a signal from neurons, an immune molecule called IL-33, indicating that it’s time for a new synapse to form, the study found. When researchers used genetic tools to block this signal, microglia failed to fulfill their ECM-chomping duties, leading to fewer new connections between neurons in the brain of mice and leaving mice struggling to remember certain details over time. When researchers instead drove the level of IL-33 signaling up, new synapses increased in number. In older mice, in which brain aging already slows the formation of new connections, ramping up IL-33 helped push the number of new synapses towards a more youthful level.
The study could be important for understanding – and perhaps one day treating – the kinds of memory problems we see in age related diseases like Alzheimer’s, according to study co-author Mazen Kheirbek, PhD, an associate professor of psychiatry whose lab studies brain circuits involved in mood and emotion. But the findings might also be important for specific types of emotional memory problems sometimes seen in anxiety related disorders.
To determine how changes in IL-33 affect memory, the researchers taught mice to distinguish between an anxiety-inducing box (inside which the mice received a mild foot shock) and a neutral box. After a month, normal mice expressed far more fear in the shock-associated box by freezing in place (a rodent reflex to throw off predators) than they did in the neutral box, where they moved around more casually. But mice with disrupted IL-33 expressed high levels of fear in either box, suggesting they’d lost the kind of precise memory needed to determine when they should be scared and when they were safe.
Kheirbek likens this overgeneralized response to the kind of trauma-induced fear that might result from being mugged in a parking lot at night. Instead of being able to separate that fearful memory from new, perhaps less-threatening experiences, some people might develop a generalized fear that makes it hard for them to enter any parking lot at any time. “Deficits in this ability to have very precise, emotional memories are seen in a lot of anxiety disorders and particularly in PTSD,” he said. “It’s an overgeneralization of fear that can really interfere with your life.”
For Molofsky’s part, stumbling upon this unexpected finding has left her eager to learn more about the ECM and how it shapes the way we learn. Her lab is now working to identify new, poorly characterized pieces of the matrix to look for as yet undocumented ways it interacts with neurons and microglia in the brain.
“I’m in love with the extracellular matrix,” Molofsky said. “A lot of people don’t realize that the brain is made up not just of nerve cells, but also cells that keep the brain healthy, and even the space in between cells is packed with fascinating interactions. I think a lot of new treatments for brain disorders can come from remembering that.”
Authors: Anna Molofsky is the senior and corresponding author on the study, and Phi Nguyen is the study’s lead author. Other authors were Leah Dorman, Simon Pan, Ilia Vainchtein, Rafael Han, Hiromi Nakao-Inoue, Sunrae Taloma, Jerika Barron, Ari Molofsky, and Mazen Kheirbek, all at UCSF.
Funding: The research was supported by the Pew Charitable Trusts, the National Institute of Mental Health (R01MH119349, DP2MH116507, R01 MH108623, R01 MH111754, R01 MH117961), the National Science Foundation (graduate research fellowship #1650113), the Burroughs Wellcome Fund, a Weill Scholar Award, the Esther A. and Joseph Klingenstein Fund, and a One Mind Rising Star Award.
Disclosures: The authors declare no competing interests.
The University of California, San Francisco (UCSF) is exclusively focused on the health sciences and is dedicated to promoting health worldwide through advanced biomedical research, graduate-level education in the life sciences and health professions, and excellence in patient care. UCSF Health, which serves as UCSF’s primary academic medical center, includes top-ranked specialty hospitals and other clinical programs, and has affiliations throughout the Bay Area.