Researchers have developed a low-cost, simple imaging system that uses tumor-targeting fluorescent molecules to determine the depth of tumor cells in the body. The portable system could eventually help surgeons distinguish between healthy and cancerous tissue with higher precision when removing a tumor.
Doctors can use fluorescent molecules during tumor resection to make cancer cells light up so that the surgeon can see if any cancerous tissue remains. However, the equipment necessary for this technique isn’t widely accessible and typically doesn’t provide quantitative information about how deep within the tissue the cancer cells reside. Having access to depth information would help surgeons to remove a full healthy layer of tissue around the tumor, which has been shown to provide the best possible outcomes for patients.
“The few commercial systems that do provide quantitative depth information are large and expensive, limiting use outside of large medical centers,” said research team leader Christine M. O’Brien from Samuel Achilefu’s Lab at Washington University School of Medicine in St. Louis. “Our group built upon prior work in this field to develop a low-cost, simple system that can quickly determine the depth of tumor cells using near-infrared (NIR) fluorescent probes.”
The researchers describe their new system in the Optica Publishing Group journal Biomedical Optics Express. The portable and easy-to-use system could be used in low-resource clinical centers, which could help minimize health disparities.
“Systems like this could be used in the future to improve surgical outcomes of patients undergoing tumor removal,” said O’Brien. “It would also prevent the need to wait for pathology results before confirming whether cancer cells are still present after tumor removal.”
Lighting up cancer
Research has shown that surgical treatments for cancer tend to be most successful if surgeons remove not only the tumor but also a healthy layer of tissue completely surrounding it. However, this can be difficult because it’s hard to pinpoint the margins between where the tumor ends and healthy tissue begins. In addition, the optimal thickness of the healthy layer depends on the tumor type and location.
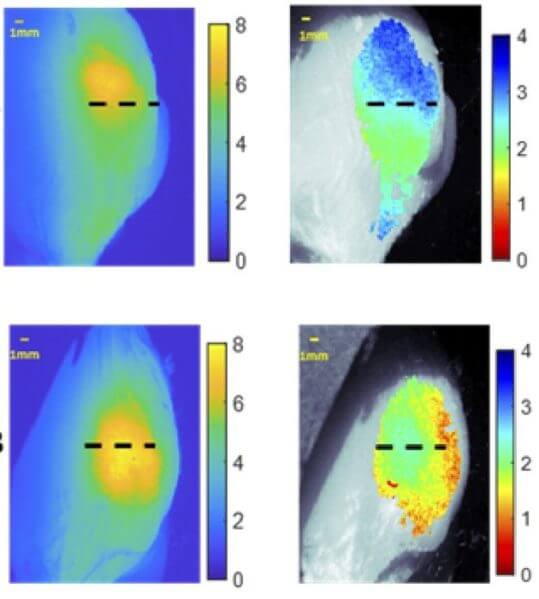
To help with this task, the Achilefu Lab research team led by O’Brien developed a new instrument based on applying one single fluorescent dye during tumor resection which then can be excited by two different NIR-wavelengths that penetrate different depths in the tissue. The emitted NIR-fluorescence can be imaged through tissue, allowing cancer cells to be detected 1 to 2 centimeters below the surface.
Dual wavelength excitation fluorescence uses the fact that different colors, or wavelengths, of light will travel different distances within tissue. By illuminating tumor-targeting fluorescent molecules with different light wavelengths and comparing their responses, it’s possible to predict how deep the tumor-targeted agents are located within the tissue.
“Multiple research groups have contributed to the development of mathematical relationships that link fluorophore depth to ratiometric fluorescence measurements,” said O’Brien. “The surge of near-infrared contrast agents being developed for use in medicine encouraged us to build upon prior work and to create a system that works in the near-infrared and that is also low-cost and simple to use.”
Building a dual-wavelength system
The new fluorescence imaging system uses 730-nm and 780-nm LEDs to provide the two wavelengths of excitation light and a monochrome CMOS camera to detect the resulting fluorescence. An 850-nm LED was also incorporated to create a brightfield image that allows the fluorescence images to be correlated with the real-world view of the tissue. The researchers decided to use an experimental agent called LS301, which can be administered during tumor resection, developed in the Achilefu Lab, as the cancer-targeting infrared probe because its broad excitation spectrum prevents the need to use more than one fluorophore, which would have made the clinical application more complex. LS301 is currently undergoing clinical trials in breast cancer patients.
After testing the system on layered synthetic materials and slices of chicken, the researchers assessed its ability to predict the depth of a tumor from breast tumors grown in mice. This was done by injecting the mice with LS301 and then imaging them using the system. Capturing the necessary images took 5 minutes. Calculations based on these images correlated well with the true depth of the tumor, showing an average error of only 0.34 mm, which is likely acceptable for clinical use.
The researchers are now working to make the system even more useful for surgical guidance by speeding up data processing and adding additional automation to the system so that it can scan the entire tissue surface.
Paper: C. M. O’Brien, K. Bishop, H. Zhang, X. Xu, L. Shmuylovich, E. Conley, K. Nwosu, K. Duncan, S. B. Mondal, G. Sudlow, S. Achilefu, “Quantitative tumor depth determination using dual wavelength excitation fluorescence,” Biomed. Opt. Express, volume 13, issue 1qq, pp. 5628-5642 (2022).
DOI: https://doi.org/10.1364/BOE.468059