B cells or B lymphocytes are an important part of the body’s immune system. When healthy B cells are infected with the Epstein-Barr virus (EBV), they undergo growth transformation, a process that immortalizes B cells, leading to their uncontrolled proliferation.
The induction of immortalized B cells is the first step in the development of posttransplant lymphoproliferative disorder (PTLD), which can evolve to lymphoma and other lymphoproliferative disorders. One key feature of B cell growth transformation is the enlargement of cells and nuclei. However, the big question remains—what are the exact molecular mechanisms regulating EBV-induced B cell growth transformation?
A recent study by Japanese researchers, published online in Microbiology Spectrum on 6 July 2023, has now provided the answer. By using primary B cells from healthy donors instead of cell lines, the team uncovered the genetic machinery responsible for inducing growth transformation following EBV infection. Explaining the rationale behind the study, principal investigator Prof. Takayuki Murata commented, “Insights from EBV research that uses cell lines has been limited, because cell lines are already in an immortalized state. To overcome this roadblock, we used primary B cells from healthy donors and then infected them with EBV. This allowed us to monitor the step-by-step growth transformation of B cells and analyze the mechanisms involved.”
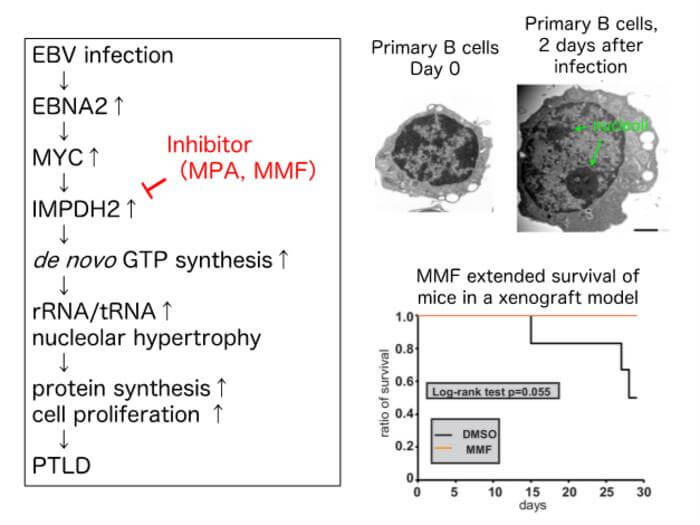
The first step involved the careful observation of primary B cells infected with wild-type EBV using electron microscopy and immunostaining. As early as two days after infection, the morphology of B cells showed significant alterations. An enlargement of the nucleolus (a region within the nucleus that produces ribosomes, the cell’s protein-producing machinery) was observed, along with an increase in the number of nucleoli. Interestingly, nucleolar enlargement was followed by an enlargement of both—the nuclei and the cells.
To understand the transcriptional changes that occurred in infected B cells, RNA sequencing was performed. “Among the genes showing significantly altered expression levels, one named IMPDH2 stood out, as it had previously been linked to similar morphological changes in glioblastoma (another type of cancer). Careful analysis showed that the levels of the IMPDH2 peaked two days after infection—coinciding with the timing of nucleolar enlargement. This suggested that we were on the right path,” explained Dr. Atsuko Sugimoto from Fujita Health University School of Medicine, who was also a part of the research team.
Interestingly, changes such as IMPDH2 induction and nucleolar enlargement could be seen when primary B cells were activated using inflammatory signals, even in the absence of EBV infection. Finally, the inhibition of IMPDH2 using silencer RNAs and the drug mycophenolic acid (MPA) prevented the growth transformation of primary B cells after EBV infection, producing smaller nucleoli, nuclei, and cells. This confirmed that IMPDH2 induction played a key role in the growth transformation of EBV-infected B cells.
The next step involved understanding how EBV activates IMPDH2 expression. Three key viral genes—EBNA2 and LMP1—were tested because of their known role in EBV-induced B cell transformation. Interestingly, when EBV lacking EBNA2 was used for infection, IMPDH2 induction following primary EBV infection was blocked. This effect was not observed with LMP1 knockout. “This very clearly demonstrated that EBV induces IMPDH2 expression via EBNA2–dependent mechanisms. In addition, cellular transcription factor MYC was also involved in the IMPDH2 induction,” clarified Dr. Sugimoto.
With several key pieces of evidence on their plate, the researchers finally set out to find the final piece of the puzzle. To highlight the clinical significance of their findings, they examined whether the drug mycophenolate mofetil (MMF) could prevent B cell transformation and PTLD. Prof. Murata elaborated, “Like MPA, which we tested in the earlier part of our study, MMF is an IMPDH2 inhibitor. More importantly, MMF is already a clinically approved immunosuppressant. That is why it was useful to test if it could be applied for the clinical prevention of PTLD.” As expected, the administration of MMF in a pre-clinical mouse xenograft model led to improved survival and reduced splenomegaly (enlargement of the spleen, indicating reduced B cell proliferation). These observations confirmed that the use of MMF can inhibit PTLD development.
This study is the first to demonstrate that IMPDH2 activation and nucleolar hypertrophy are essential for EBV-induced B cell transformation and that IMPDH2 inhibition can suppress PTLD. It could lead to the adoption of MMF as an agent for the prevention of EBV-positive PTLD, providing significant relief to transplant patients.