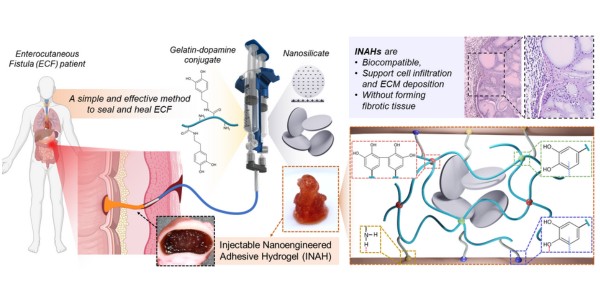
In a major advancement in medical technology, researchers at the Terasaki Institute for Biomedical Innovation have developed an adhesive gel, offering a revolutionary treatment for gastrointestinal leaks, a condition clinically known as enterocutaneous fistulas. This development marks a significant milestone in addressing a challenging medical condition that has long plagued patients and clinicians.
Enterocutaneous fistulas, which are abnormal connections between the gastrointestinal tract and the skin, are primarily a consequence of surgical complications and cause intestinal and stomach contents to leak through to the skin. This can lead to severe health risks, including skin breakdown, increased risk of infection, and substantial mortality rates estimated between 15 and 20%. The current definitive treatment usually requires major surgical intervention, which aims for refunctionalization of the entire bowel, removal of the fistula with end-to-end reconnection of the bowel, and secure abdominal wall closure. Hence, there is an unmet need for minimally invasive approaches to seal and heal these gastrointestinal leaks.
The adhesive gel represents a paradigm shift in the treatment of gastrointestinal leaks. As outlined in their paper published in Acta Biomaterialia, the gel is composed of synthetic nanoparticles and gelatin modified with dopamine; this gel is designed to quickly solidify upon injection, forming a robust structure with superior mechanical and adhesive properties. This unique composition ensures that the gel can effectively seal the fistula, preventing further complications and aiding in healing.
One of the most notable features of the adhesive gel is its easy injection and formation of a stable structure that supports tissue regeneration and integrates seamlessly with surrounding tissues. This property is crucial in medical applications where precision and minimally invasive procedures are paramount.
In addition to its structural benefits, the adhesive gel has shown impressive biological compatibility. Tests with animals have demonstrated that the gel supports cell infiltration and deposition of proteins essential for new tissue growth, crucial steps in the healing process. Moreover, it achieves these results without forming fibrotic tissue, a common problem in many tissue repair strategies.
The adhesive properties of the gel have been tested with skin and artery models, where it displayed remarkable volume stability and sustained strong adhesion in hydrated internal environments. This characteristic is vital for maintaining the integrity of the fistula repair over time.
Furthermore, the gel’s potential for localized drug delivery represents a significant advancement in medical treatment. By incorporating bioactive cues, the adhesive gel can guide cell behavior and promote functional tissue regeneration, offering a versatile solution for sealing and healing gastrointestinal leaks and broader applications in tissue regeneration and drug delivery systems.
This research presents a beacon of hope for patients suffering from gastrointestinal leaks. The development of the adhesive gel stands as a testament to the power of innovative biomedical research and its potential to transform patient care. It offers a more straightforward, more effective method for treating a condition that has long been a challenge in the medical community, paving the way for improved outcomes and enhanced quality of life for patients.
As we look to the future, the implications of this research extend beyond the immediate treatment of gastrointestinal leaks. The principles and technologies developed here could have far-reaching effects in tissue engineering and regenerative medicine, opening new avenues for research and treatment in various medical conditions.
In conclusion, the adhesive gel developed by the Terasaki Institute for Biomedical Innovation represents a significant leap forward in medical science. Its unique properties and promising results highlight its potential as a game-changing solution for treating gastrointestinal leaks and possibly other medical conditions needing effective tissue repair and regeneration strategies.
Authors: Natan Roberto de Barros, Ankit Gangrade, Ahmad Elsebahy, RunRun Chen, Fatemeh Zehtabi, Menekse Ermis, Natashya Falcone, Reihaneh Haghniaz, Safoora Khosravi, Alejandro Gomez, Shuyi Huang, Marvin Mecwan, Danial Khorsandi, Junmin Lee, Yangzhi Zhu, Bingbing Li, HanJun Kim, Finosh G Thankam, Ali Khademhosseini.
The authors acknowledge funding from the National Institutes of Health (1R01DK130566-01, 1R01CA257558-01) and the Terasaki Institute for Biomedical Innovation, Los Angeles, CA. H-J.K. would like to acknowledge the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (RS-2023-00240729). This research was supported by the MSIT (Ministry of Science and ICT), Korea, under the ITRC (Information Technology Research Center) support program (IITP-2023-RS-2023-00258971) supervised by the IITP (Institute for Information & Communications Technology Planning & Evaluation). This work was also supported by a Korea University Grant (K2326671). B. Li and his students are sponsored by the NASA MUREP Institutional Research Opportunity (MIRO) grant (80NSSC19M0200) and MUREP High Volume grant (80NSSC22M0132).