As direct descendants of ancient bacteria, mitochondria have always been a little alien.
Now a study shows that mitochondria are possibly even stranger than we thought.
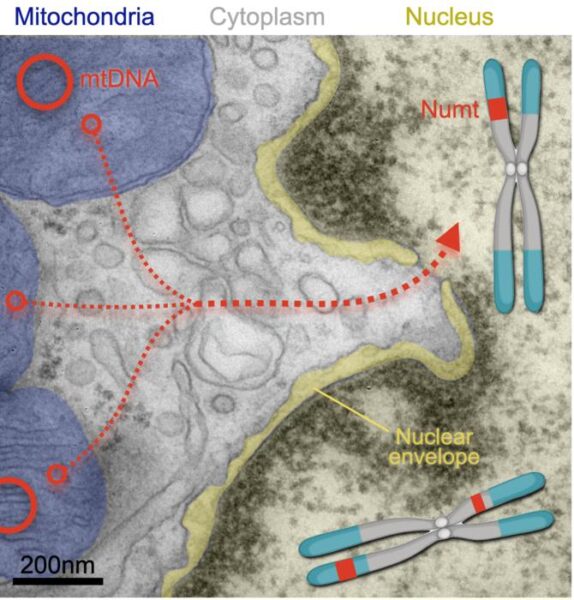
Mitochondria in our brain cells frequently fling their DNA into the nucleus, the study found, where the DNA becomes integrated into the cells’ chromosomes. And these insertions may be causing harm: Among the study’s nearly 1,200 participants, those with more mitochondrial DNA insertions in their brain cells were more likely to die earlier than those with fewer insertions.
“We used to think that the transfer of DNA from mitochondria to the human genome was a rare occurrence,” says Martin Picard, mitochondrial psychobiologist and associate professor of behavioral medicine at Columbia University Vagelos College of Physicians and Surgeons and in the Robert N. Butler Columbia Aging Center. Picard led the study with Ryan Mills of the University of Michigan.
“It’s stunning that it appears to be happening several times during a person’s lifetime, Picard adds. “We found lots of these insertions across different brain regions, but not in blood cells, explaining why dozens of earlier studies analyzing blood DNA missed this phenomenon.”
Mitochondrial DNA behaves like a virus
Mitochondria live inside all our cells, but unlike other organelles, mitochondria have their own DNA, a small circular strand with about three dozen genes. Mitochondrial DNA is a remnant from the organelle’s forebears: ancient bacteria that settled inside our single-celled ancestors about 1.5 billion years ago.
In the past few decades, researchers discovered that mitochondrial DNA has occasionally “jumped” out of the organelle and into human chromosomes.
“The mitochondrial DNA behaves similar to a virus in that it makes use of cuts in the genome and pastes itself in, or like jumping genes known as retrotransposons that move around the human genome,” says Mills.
The insertions are called nuclear-mitochondrial segments—NUMTs (“pronounced new-mites”)—and have been accumulating in our chromosomes for millions of years.
“As a result, all of us are walking around with hundreds of vestigial, mostly benign, mitochondrial DNA segments in our chromosomes that we inherited from our ancestors,” Mills says.
Mitochondrial DNA insertions are common in the human brain
Research in just the past few years has shown that “NUMTogenesis” is still happening today.
“Jumping mitochondrial DNA is not something that only happened in the distant past,” says Kalpita Karan, a postdoc in the Picard lab who conducted the research with Weichen Zhou, a research investigator in the Mills lab. “It’s rare, but a new NUMT becomes integrated into the human genome about once in every 4,000 births. This is one of many ways, conserved from yeast to humans, by which mitochondria talk to nuclear genes.”
The realization that new inherited NUMTs are still being created made Picard and Mills wonder if NUMTs could also arise in brain cells during our lifespan.
“Inherited NUMTs are mostly benign, probably because they arise early in development and the harmful ones are weeded out,” says Zhou. But if a piece of mitochondrial DNA inserts itself within a gene or regulatory region, it could have important consequences on that person’s health or lifespan. Neurons may be particularly susceptible to damage caused by NUMTs because when a neuron is damaged, the brain does not usually make a new brain cell to take its place.
To examine the extent and impact of new NUMTs in the brain, the team worked with Hans Klein, assistant professor in the Center for Translational and Computational Neuroimmunology at Columbia, who had access to DNA sequences from participants in the ROSMAP aging study (led by David Bennett at Rush University). The researchers looked for NUMTs in different regions of the brain using banked tissue samples from more than 1,000 older adults.
Their analysis showed that nuclear mitochondrial DNA insertion happens in the human brain—mostly in the prefrontal cortex—and likely several times over during a person’s lifespan.
They also found that people with more NUMTs in their prefrontal cortex died earlier than individuals with fewer NUMTs. “This suggests for the first time that NUMTs may have functional consequences and possibly influence lifespan,” Picard says. “NUMT accumulation can be added to the list of genome instability mechanisms that may contribute to aging, functional decline, and lifespan.”
Stress accelerates NUMTogenesis
What causes NUMTs in the brain, and why do some regions accumulate more than others?
To get some clues, the researchers looked at a population of human skin cells that can be cultured and aged in a dish over several months, enabling exceptional longitudinal “lifespan” studies.
These cultured cells gradually accumulated several NUMTs per month, and when the cells’ mitochondria were dysfunctional from stress, the cells accumulated NUMTs four to five times more rapidly.
“This shows a new way by which stress can affect the biology of our cells,” Karan says. “Stress makes mitochondria more likely to release pieces of their DNA and these pieces can then ‘infect’ the nuclear genome,” Zhou adds. It’s just one way mitochondria shape our health beyond energy production.
“Mitochondria are cellular processors and a mighty signaling platform,” Picard says. “We knew they can control which genes are turned on or off. Now we know mitochondria can even change the nuclear DNA sequence itself.”
Additional information
The study, titled “Somatic nuclear mitochondrial DNA insertions are prevalent in the human brain and accumulate over time in fibroblasts,” was published Aug. 22 in PLOS Biology.
All authors: Weichen Zhou (University of Michigan), Kalpita R. Karan (Columbia), Wenjin Gu (Michigan), Hans-Ulrich Klein (Columbia), Gabriel Sturm (Columbia and University of California, San Francisco), Philip L. De Jager (Columbia), David A. Bennett (Rush University Medical Center), Michio Hirano (Columbia), Martin Picard (Columbia), and Ryan E Mills (Michigan).
This work was supported by grants from the U.S. National Institutes of Health (R01AG066828, R21HG011493, and P30AG072931), the Baszucki Brain Research Fund, and the University of Michigan Alzheimer’s Disease Center Berger Endowment.
The authors report no conflicts of interest.