Fighting cancer is exhausting for T cells. Hostile tumor microenvironments can drain their mitochondrial activity, leading to a condition known as T cell exhaustion. This phenomenon also hinders adoptive cell therapies, in which healthy, tumor-targeting T cells are infused into patients with cancer. A novel method to boost mitochondrial activity and charge up T cells is needed.
Investigators from Brigham and Women’s Hospital, a founding member of the Mass General Brigham healthcare system, in collaboration with colleagues at Leibniz Institute for Immunotherapy in Germany, have developed a way to “supercharge” T cells by supplying them with extra mitochondria from multipotent stromal cells. In a new study published in Cell, researchers report that these supercharged T cells exhibited heightened anti-tumor activity and reduced signs of exhaustion in preclinical models of cancer, suggesting that this technique could help improve existing immunotherapies.
“These supercharged T cells overcome one of the fundamental barriers of immunotherapy by penetrating the tumor and overcoming immune barren state in the tumor,” said corresponding author Shiladitya Sengupta, PhD, of the Brigham’s Department of Medicine. “Mitochondria provide the fuel. It’s like we’re taking T cells to the fuel station and gassing them up. This transplanting of mitochondria is the dawn of organellar therapy where an organelle is delivered to a cell to make it more effective.”
“Previous efforts to enhance mitochondrial function in T cells have focused on targeting specific genes or pathways, but these methods fall short when the mitochondria are already damaged or dysfunctional. Our approach involves the transfer of whole, healthy mitochondria organelles into the cells. This process is comparable to organ transplantation—like heart, liver, or kidney transplants—but conducted at a microscopic level,” explained Luca Gattinoni, MD, co-leading author of the study.
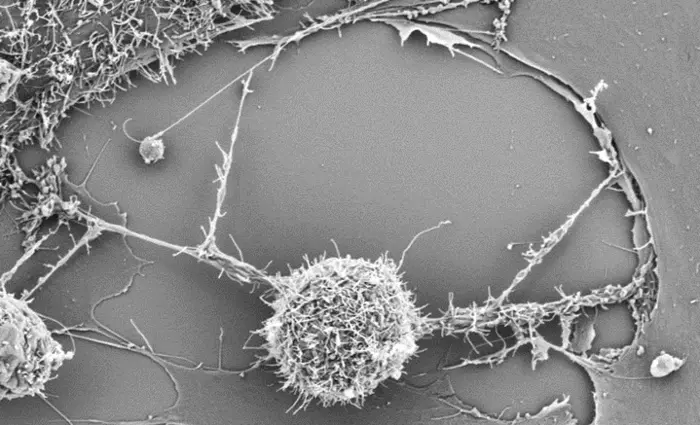
To develop this method, the researchers built upon their previous findings, which showed that cancer cells can slurp up mitochondria from immune cells through intercellular nanotubes that the researchers have described as “tiny tentacles.” Drawing from these results, the group teamed up with scientists at the Leibniz Institute to investigate interactions between bone marrow stromal cells (BMSCs) and cytotoxic T cells. Using varied electron and fluorescent microscopy approaches, they observed that BMSCs extended nanotubes to activated T cells, donating intact mitochondria. This helped to revive the T cells (mito+), which showed increased respiratory capacity, a sign of enhanced metabolism.
The research team examined how supercharging the T cells affected immune function. When infused into a mouse model of melanoma, mito+ cells showed significantly higher anti-tumor responses and prolonged survival rates compared to T cells without additional mitochondria. Further experiments revealed that mito+ cells could easily penetrate tumors, multiply quickly and pass on their extra mitochondria to daughter cells, where they persisted for a long time. In addition, the authors discovered that mito+ cells could survive and resist T cell exhaustion within the tumor microenvironment.
The researchers found that supercharging human T cells helped the immune system fight tumors in multiple models of cancer. Of note, they demonstrated that tumor-infiltrating lymphocytes and CAR-Ts, which often develop damaged mitochondria within the tumor microenvironment, displayed augmented cancer-destroying properties when boosted with mitochondria from primary BMSCs from human donors.
The authors suggest that future applications could include using patient-matched BMSCs to supercharge T cells for adoptive transfer.
Authorship: In addition to Gupta, BWH authors include Tanmoy Saha. Additional authors include Jeremy G. Baldwin, Christoph Heuser-Loy, Roland Schelker, Dragana Slavkovic-Lukic, Nicholas Strieder, Inmaculada Hernandez-Lopez, Nisha Rana, Markus Barden, Fabio Mastrogiovanni, Azucena Martín-Santos, Andrea Raimondi, Philip Brohawn, Brandon W. Higgs, Claudia Gebhard, Veena Kapoor, William G. Telford, Sanjivan Gautam, Maria Xydia, Philipp Beckhove, Sina Frischholz, Kilian Schober, Zacharias Kontarakis, Matteo Iannacone, Jessica Fioravanti.
Disclosures: Baldwin, Saha, Fioravanti, Sengupta, and Gattinoni have a patent application for the use of mitochondrial transfer technology in cancer immunotherapies. Brohawn and Gautam have an employee relationship and have stock in AstraZeneca. Higgs has an employee relationship and has stock in Genmab. Gattinoni has consulting agreements with Lyell Immunopharma, Instil Bio, and Advaxis. Gattinoni is on the scientific advisory board of Poseida Therapeutics and Kiromic and a stockholder of Poseida Therapeutics. Iannocone participates in advisory boards/consultancies for Gilead Sciences, Third Rock Ventures, Antios Therapeutics, Asher Biotherapeutics, GentiBio, Clexio Biosciences, Sybilla, and BlueJay Therapeutics. Fioravanti has an employee relationship and has stock in Lyell Immunopharma. Sengupta is a founder and owns equity in Vyome Therapeutics Inc. and Alyssum Therapeutics Inc.
Funding: This research was supported by the NIH Intramural Research Program through an NCI FLEX award, the CRI Clinic and Laboratory Integration Program (CLIP) (CRI3201), NIH-NCI RO1s RO1CA293908 and RO1CA276525 and the DFG Reinhart Koselleck project (GA 2882/2-1).
Paper cited: Baldwin JG et al. “Intercellular nanotube-mediated mitochondrial transfer enhances T cell metabolic fitness and antitumor efficacy” Cell DOI: 10.1016/j.cell.2024.08.029