Adult stem cells represent a sort of blank clay from which a myriad of different cell and tissue types are molded and as such are of critical importance to health, ageing and disease. In tissues that turn over rapidly, such as the intestines, the self-renewing nature of stem cells and their susceptibility to cancer-causing mutations has led researchers to postulate that these cells also act as the cell of origin in cancers. The rarity of adult stem cells relative to their differentiated daughter cells has, however, made them historically difficult to study.
Over the years, researchers have hypothesized that the body maintains a population of mutation- and injury-resistant “reserve” stems cells that serve as a kind of dormant reservoir from which all other cells in a given tissue can be derived. Yet researchers have been conflicted about the precise identity of this population of cells.
Now, a team from the University of Pennsylvania has helped identify key characteristics that distinguish reserve stem cells from other stem cell populations that had been purported to have similar properties. The work, which employed single-cell gene expression analyses as well as other cutting-edge techniques, demonstrated that, in the intestines, reserve stem cells are a distinct population from so-called “label-retaining cells.” The two populations were long believed to be one and the same.
“The devil is in the details,” said senior author Christopher J. Lengner, an assistant professor in the Department of Biomedical Sciences in Penn’s School of Veterinary Medicine and member of the Penn Institute for Regenerative Medicine. “You need an assay with single-cell sensitivity to address the potential heterogeneity in the cell population being study and thus to truly understand what these cells are. Now that we have that level of resolution, we can begin to ask questions that are relevant to questions such as how cancer is initiated, a process that starts in a single cell.”
The paper appears in the journal Gastroenterology.
Lengner collaborated on the work with Ning Li, the paper’s lead author, and Angela Nakauka-Ddamba, both of Penn Vet; John Tobias of the Penn Genome Analysis Core; and Shane T. Jensen of Penn’s Wharton School.
The concept of the reserve stem cell arose in the mid-1970s, when biologist John Cairns formulated a theory that became known as the immortal strand hypothesis. His idea was that reserve stem cells remained immune from mutation by protecting the “mother strand” of DNA. During each cell division, the theory went, only the newly replicated strands of DNA were passed to the daughter cell, while the mother strand was maintained by the original stem cell.
In search of evidence to test this hypothesis, researchers conducted experiments in which they coaxed stem cells to incorporate a radioactive DNA label and then tracked it through cell divisions.
“What they saw then was after two months there were still rare cells that had retained this label,” Lengner said. “They took this and some other experiments as evidence for the immortal strand hypothesis.”
In other words, they believed that the cells that retained the label were the reserve stem cells that had held onto the mother strand of DNA.
A few decades later, the immortal strand hypothesis lost favor due to a lack of supporting evidence, but scientists explained the existence of the so-called label-retaining cells by noting that they could be a population of stem cells that divided only rarely. In the early 2000s, researchers used different labels, including green fluorescent protein, or GFP, fused to histone proteins that are stably incorporated into the chromatin, to track cell division. Using GFP had the added benefit of allowing researchers to prospectively isolate living cells using fluorescence-activated cell sorting and to subsequently analyze cell behavior and gene expression in the purified cell populations.
These types of experiments have been used even more recently to suggest that label-retaining cells are the same as reserve stem cells. These recent studies have indicated that at least some cells within the label-retaining population could give rise to all the different cell lineages in the intestine, just as a stem cell ought to.
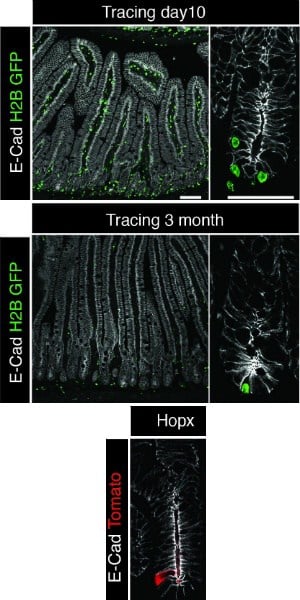
The Penn team, however, decided to take a closer look at the label-retaining cells to see if they were truly the same thing as the indispensable reserve stem cells. To do so, they created an experimental system whereby they introduced GFP into young mice targeted to intestinal stem cells and then monitored the loss of fluorescence over three months. As observed previously, they found cells that retained the GFP label. Over the short term, these label-retaining cells were at and above the base of the intestinal crypt, an invagination at the base of the intestinal villi protected from the contents of the lumen. As more time passed, over one and then three months, the cells were found more rarely and became localized exclusively at the very base of the crypt.
“This was one piece of data that confused people,” Lengner said, “because label-retaining cells and reserve stem cells can both be found roughly at the same position relative to the crypt base. This led people to speculate that they were one in the same.”
To establish whether they were indeed the same, the Penn team bred two populations of mice together, one with the GFP marking label-retaining cells and another with reserve intestinal stem cells labeled with a genetic marker controlled by the Hopx gene in red. In the resulting offspring, they found a few cells with both labels, but the majority of labeled cells fell into mutually exclusive populations.
The researchers then used single-cell gene expression profiling to look for genes in the different cell populations. They found that the short-term label-retaining cells, reserve intestinal stem cells and a cell population known as active crypt base columnar intestinal stem cells, which are known to be susceptible to injury and mutation, each had distinctive gene expression profiles and were thus distinct populations.
Subjecting the various cell populations to radiation injury, they found that the majority of the Hopx-marked cells, or reserve intestinal stem cells, survived and entered the cell cycle to regenerate the damaged tissue, while short-term label-retaining cells did not. These cell populations had additional functional differences. While the majority of Hopx-marked stem cells were in the resting, or quiescent stage of the cell cycle, most label-retaining cells were arrested in the G1 phase of the cell cycle, characteristic of differentiated cells.
Finally, the researchers showed that Hopx-marked intestinal stem cells could form intestinal organoids in culture while long-term label-retaining cells could not, indicating that the long-term label-retaining cells were already terminally differentiated and could not revert back. Short-term label-retaining cells did form organoids but only about half as efficiently as the Hopx cells did.
“The conclusion we come to is that these short-term label-retaining cells are progeny of active stem cells and are undergoing a bifurcation to be one of two types of intestinal cells,” Lengner said. “They retain some ability to revert to the stem cell state in culture, but they’re not the radiation-resistant reserve stem cell that we and other groups have long studied.”
The findings, which challenge decades of previous work, provide researchers with a new framework with which to study intestinal stem cells.
“It’s like you’re watching a soccer game and the players all have numbers on their backs,” said Li. “If they didn’t have those numbers, it would be very difficult to talk about the game and know whether the player I’m talking about is the same player you’re talking about. It’s the same with these cell populations.”
Now that they’ve established that reserve stem cells are distinct from the label-retaining cells, the Penn researchers would like to put more emphasis on studying the true reserve stem cells to determine how cell populations in the intestine are organized and, eventually, perhaps to understand how these and other cell populations figure in the emergence of cancer.
The study was supported by the National Cancer Institute and National Institute of Diabetes and Digestive and Kidney Disorders.
If our reporting has informed or inspired you, please consider making a donation. Every contribution, no matter the size, empowers us to continue delivering accurate, engaging, and trustworthy science and medical news. Independent journalism requires time, effort, and resources—your support ensures we can keep uncovering the stories that matter most to you.
Join us in making knowledge accessible and impactful. Thank you for standing with us!