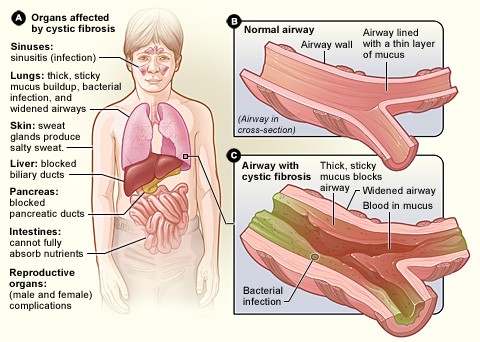
Two new studies from the University of Iowa suggest that gene therapy may be a viable approach for treating or preventing lung disease caused by cystic fibrosis (CF).
Working with CF pigs, the researchers, based in the UI Pappajohn Biomedical Institute (PBI), have shown that two different virus-based vectors can restore a working version of a protein—the cystic fibrosis transmembrane conductance regulator (CFTR)—that is faulty in CF to the pigs’ airway cells. Moreover, this gene replacement normalizes important aspects of the lung biology and improves the ability of airway secretions to kill bacteria.
“This is an important proof of principle for the idea that gene therapy for CF could work because we used an animal model that we know develops lung disease like people,” says Paul McCray Jr., professor of pediatrics in the UI Carver College of Medicine and principal investigator for one of the studies, both of which were published Sept. 8 in the journal JCI Insight. “In our short-term experiments we saw evidence of correction of some of the known problems of CF, including salt movement across the cell membrane, the pH (acidity) of the airway surface liquid, and the ability of respiratory secretions to kill bacteria. This shows that the gene therapy has an effect that appears to be therapeutically relevant.”
Despite the simplicity of the gene therapy concept—replacing a disease-causing gene with a normal version—safely delivering genes to the correct cells so that they produce sufficient amounts of the replacement protein to treat a disease has proved difficult to achieve.
For CF, one significant hurdle has been the lack of a good animal model to test the therapeutic outcomes of gene therapies. Mice with CF do not develop the lung disease that is the major cause of illness and death in human patients.
In 2008, CF researchers at the UI led by Michael Welsh, director of the PBI and a Howard Hughes Medical Institute investigator, made a major advance by creating a pig model of CF, which, unlike CF mice, develops lung disease that mimics the symptoms seen in people.
“The pig model allows us to look at the effect of gene therapy on lung disease and ask whether the gene rescue also improves the lung problems caused by CF,” says Joseph Zabner, UI professor of internal medicine.
In the new studies, two teams tested two different gene therapy strategies to get functional CFTR into the airway cells of CF pigs. One group, led by McCray and Patrick Sinn, UI research associate professor of pediatrics and director of the UI Viral Vector Core, focused on a lentivirus. This type of virus has been successfully and safely used as a gene therapy vector for patients with rare immune diseases. A major advantage of lentiviruses is the delivered gene is directly incorporated (integrated) in to the cell’s genome, meaning the fix is permanent. However, it is challenging to produce large quantities of lentivirus, and this virus has not yet been tested for safety in human lungs.
The other research team, led by Zabner and David Schaffer at the University of California, Berkeley, focused on the adeno-associated virus AAV2. AAVs are safe for use in humans, including human lungs, and relatively easy to produce in large quantities. Genes delivered by AAV vectors are not permanently incorporated into the cell’s genome, but expression of the gene is often long-lived.
An important aspect of the AAV study was the molecular customization of the virus such that it efficiently targeted pig airway cells. Zabner and Schaffer created an AAV2 virus with five mutations that was 240 times more efficient than AAV2 at infecting pig airway cells. The team previously used the same directed evolution strategy to create an AAV virus that preferentially infects human airway cells.
The researchers showed that both gene therapies restored chloride currents in pig airway cells, indicating that both vectors delivered working CFTR to the correct location in the airways. Both approaches also increased the pH and the bacterial-killing ability of the pigs’ airway secretions.
“Gene therapy development has experienced ups and downs, and for CF there has been a long period with few clinical studies,” McCray notes. “But a non-viral gene therapy trial was completed recently in the UK, and we’ve also started to see successes in other single-gene diseases. With these results, and all we have learned about getting the CFTR gene into the right place, there is renewed interest in moving forward with CF gene therapy.”
Sinn adds that the new findings may move researchers closer to a “tipping point” for initiating clinical trials.
The vectors the UI researchers are developing could also have uses beyond CF gene therapy as multifaceted gene-delivery tools.
“If anyone wants to use gene editing, including CRISPR-Cas9, they need a way to deliver it to cells. Vectors like these are the key to that,” Zabner says.
When the UI team started their gene therapy work more than 20 years ago, there were no treatments available for CF. Now, several drugs are available that help patients with specific CF mutations. However, these drugs, which are expensive and are taken daily, don’t treat the majority of patients. A successful gene therapy approach would have the advantages of treating all forms of CF, regardless of the genetic mutation, and the therapy could potentially be given just once to effect a permanent treatment.
“The big questions we are asked pertain to how feasible it would be to use this approach to treat people with CF. For example, ‘How long does the gene expression last?’ and ‘Does it prevent disease from happening?’ Those questions will need longer-term studies to answer,” says McCray.
The current studies resulted from highly collaborative science among teams from the UI, the University of California, Berkeley, and Wright State University in Ohio, as well as among multiple departments within the UI Carver College of Medicine. The two lead authors of the studies are both UI graduate students: Ashley Cooney, in the microbiology program, and Benjamin Steines, in the molecular and cellular biology program. The research was funded in part by grants from the National Heart, Lung, and Blood Institute and the National Institute for Diabetes and Digestive and Kidney Diseases, both part of the National Institutes of Health, and the Cystic Fibrosis Foundation.
If our reporting has informed or inspired you, please consider making a donation. Every contribution, no matter the size, empowers us to continue delivering accurate, engaging, and trustworthy science and medical news. Independent journalism requires time, effort, and resources—your support ensures we can keep uncovering the stories that matter most to you.
Join us in making knowledge accessible and impactful. Thank you for standing with us!