Researchers at the University of Wisconsin–Madison have developed a method combining sticky nanoparticles with high-precision protein measurement to capture and analyze a common marker of heart disease to reveal details that were previously inaccessible.
The new method, a system known as nanoproteomics, effectively captures and measures various forms of the protein cardiac troponin I, or cTnI, a biomarker of heart damage currently used to help diagnose heart attacks and other heart diseases. An effective test of cTnI variations could one day provide doctors with a better ability to diagnose heart disease, the leading cause of death in the U.S.

Ying Ge
UW–Madison Professor of Cell and Regenerative Biology and Chemistry Ying Ge, Professor of Chemistry Song Jin and chemistry graduate students Timothy Tiambeng and David Roberts led the work, which was published Aug. 6 in the journal Nature Communications. The researchers now plan to use their new method to associate the various forms of cTnI with specific heart diseases as a step toward developing a new diagnostic test.
Doctors currently use an antibody-based test called ELISA to help diagnose heart attacks based on elevated levels of cTnI in the patient’s blood sample. While the ELISA test is sensitive, patients can have high levels of cTnI in the blood without having heart disease, which can lead to expensive and unnecessary treatments for patients.

Song Jin
“So we want to use our nanoproteomics system to look into more details at various modified forms of this protein rather than just measuring its concentration,” says Ge, who is also director of the Human Proteomics Program in the UW School of Medicine and Public Health. “That will help reveal molecular fingerprints of cTnI from each patient for precision medicine.”
Measuring low-concentration proteins in the blood like cTnI is a classic needle-in-a-haystack problem. Rare, meaningful biomarkers of disease are completely overwhelmed by common and diagnostically useless proteins in the blood. Current methods use antibodies to enrich and capture proteins in a complex sample to identify and quantify proteins. But antibodies are expensive, have batch-to-batch variations, and can generate inconsistent results.
“In the future, when ELISA shows an elevated cTnI level, your doctor might order a comprehensive nanoproteomics test to determine whether it is caused by heart disease or not, and identify different types of heart disease, for more precise treatment while avoiding unnecessary care and expense for patients.”
Song Jin
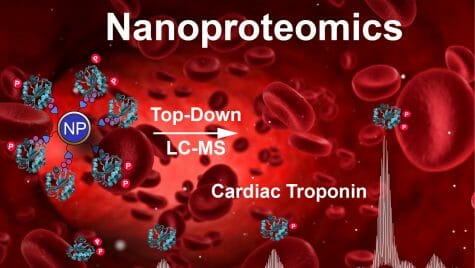
To capture cTnI and overcome some of the limitations of antibodies, the researchers designed nanoparticles of magnetite, a magnetic form of iron oxide, and linked it to a peptide of 13 amino acids long designed to specifically bind to cTnI. The peptide latches onto cTnI in a blood sample, and the nanoparticles can be collected together using a magnet. Nanoparticles and peptides are easily made in the lab, making them cheap and consistent.
Using the nanoparticles, the researchers were able to effectively enrich cTnI in samples of human heart tissue and blood. Then they used advanced mass spectrometry, which can distinguish different proteins by their mass, to not only get an accurate measurement of cTnI, but also to assess the various modified forms of the protein.
Like many proteins, cTnI can be modified by the body depending on factors like an underlying disease or changes in the environment. In the case of cTnI, the body adds various numbers of phosphate groups, small molecular tags that might change the function of cTnI. These variations are subtle and hard to track.
“But with high-resolution mass spectrometry, we can now ‘see’ these molecular details of proteins, like the hidden iceberg beneath the surface,” says Ge.
Tiambeng and Roberts decided to test if they could distinguish the various forms of cTnI that can be found in patient blood samples. They spiked blood serum with proteins from donor hearts that were normal, diseased, or from a dead donor. Then they used their nanoparticles to capture cTnI and measured the protein using mass spectrometry.
As hoped, the scientists could observe clearly different patterns in the types of cTnI prevalent in each type of heart tissue. The healthy hearts tended to have lots of cTnI with multiple phosphate groups attached, for example, while diseased hearts had cTnI that had less phosphate and the post-mortem heart had cTnI broken into pieces.
While this is still a proof-of-concept study and more research will be needed, it is this ability to associate a pattern of cTnI variations with heart health that the researchers hope could one day produce a new diagnostic tool to help when patients come to the hospital with suspected heart disease. The researchers have filed a patent application on the new technology through the Wisconsin Alumni Research Foundation.
“We like to think a future blood test based on our work here could be complementary to the current ELISA test,” says Jin. “In the future, when ELISA shows an elevated cTnI level, your doctor might order a comprehensive nanoproteomics test to determine whether it is caused by heart disease or not, and identify different types of heart disease, for more precise treatment while avoiding unnecessary care and expense for patients.”
This work was supported by the National Institutes of Health (grants R01 GM117058, R01 GM125085, R01 HL096971, S10 OD018475, T32GM008505, T32 HL007936-19).
If our reporting has informed or inspired you, please consider making a donation. Every contribution, no matter the size, empowers us to continue delivering accurate, engaging, and trustworthy science and medical news. Independent journalism requires time, effort, and resources—your support ensures we can keep uncovering the stories that matter most to you.
Join us in making knowledge accessible and impactful. Thank you for standing with us!