In a laboratory at Nanjing Agricultural University, a common soil fungus is doing something chemical engineers have struggled with for decades. Aspergillus niger, the same organism that helps ferment soy sauce and produces citric acid for fizzy drinks, is quietly dissolving phosphorus from one of the world’s most problematic industrial wastes.
The waste is phosphogypsum, and there’s rather a lot of it. Roughly 300 million tonnes accumulate globally each year as a byproduct of fertiliser production, with billions of tonnes already stacked in enormous piles that leach fluorides, radionuclides and heavy metals into surrounding soil and water. Only about 14 per cent gets reused. The rest sits there, a monument to industrial inefficiency.
Here’s the thing, though: phosphogypsum contains about 1 per cent phosphorus, locked up in insoluble forms that conventional extraction methods struggle to access economically. That might not sound like much until you consider that phosphorus is essential for all life on Earth, and we’re running through the planet’s finite reserves of phosphate rock at an alarming rate. “Phosphogypsum has long been viewed as a liability, but it actually contains a valuable nutrient that is increasingly scarce,” says Zhen Li, who led the research.
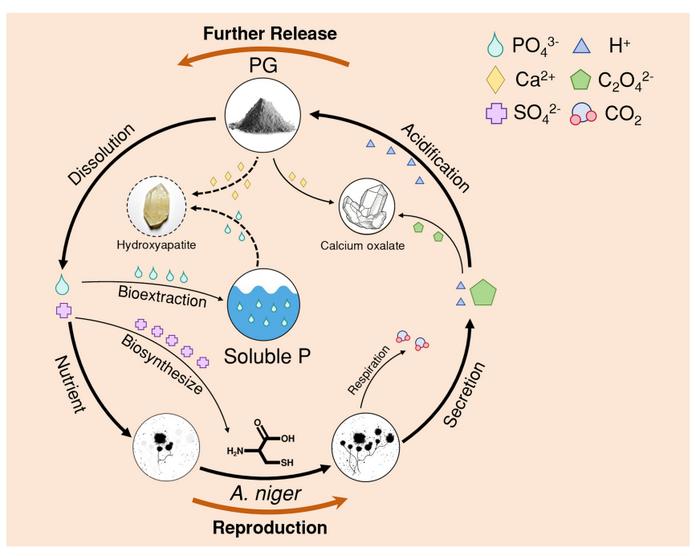
Li and his colleagues wanted to see whether biological extraction—using microorganisms instead of harsh chemicals—could recover this trapped phosphorus. They chose A. niger because it’s known for secreting substantial quantities of organic acids, particularly oxalic acid, which can dissolve phosphate minerals. The fungus also happens to be remarkably hardy, thriving in acidic conditions and high metal concentrations that would kill most organisms.
The team incubated phosphogypsum with the fungus in a modified growth medium, then watched what happened. After six days, the fungal biomass had increased to 0.85 grammes in samples with high doses of phosphogypsum, and the organisms were respiring vigorously. More importantly, phosphorus was dissolving into solution at rates far exceeding those in control samples without the fungus.
By day 15, the extraction efficiency had reached roughly 40 per cent, compared with about 10 per cent in phosphogypsum sitting in liquid without any biological help. That figure approaches the efficiency achieved when A. niger extracts phosphorus from pure calcium phosphate—impressive considering phosphogypsum is a complex, contaminated material. “Our results show that the fungus is not just dissolving phosphorus, but actively using it for growth,” Li explains. “This biological demand helps drive the extraction process forward.”
Advanced imaging using nanoscale secondary ion mass spectrometry revealed where the phosphorus was going. The researchers sliced fungal pellets into ultrathin sections and bombarded them with caesium ions, generating images showing the distribution of phosphorus, sulphur and nitrogen within individual cells. Phosphorus appeared concentrated in the fungal structures, particularly in cells undergoing division—exactly where you’d expect if the organisms were incorporating the nutrient into their own biomolecules like ATP and phospholipids.
The process works through an elegant bit of microbial chemistry. A. niger secretes oxalic acid as it grows, and the acid’s hydrogen ions create a favourable microenvironment for dissolving phosphate. Meanwhile, the oxalate component reacts with calcium in the phosphogypsum to form calcium oxalate crystals. This is crucial, because dissolved phosphorus would normally just recombine with free calcium ions to form insoluble compounds again. By tying up the calcium, the fungus prevents this re-precipitation and keeps phosphorus available in solution.
There’s a bonus, too. Phosphogypsum contains abundant sulphate, and the fungal cells were incorporating this sulphur into amino acids like cysteine. So the waste material isn’t just providing phosphorus; it’s supplying another essential nutrient that supports continued fungal growth and metabolism. Li and his team found evidence of this through the same imaging technique that revealed phosphorus uptake, with sulphur distributed throughout the cellular structures in patterns consistent with protein synthesis.
The researchers also ran computer simulations using geochemical modelling software to understand what would happen as pH changed during incubation. Without the fungal intervention, increasing pH would cause phosphate ions to combine with calcium and mineralise into hydroxylapatite, effectively locking the phosphorus away again. But the abundant oxalic acid production prevents this, maintaining conditions where phosphorus stays accessible.
It’s worth noting that the fungal hyphae—those threadlike structures that make up the organism’s body—can physically wrap around dispersed phosphogypsum particles, creating close contact that extends the reach of the organic acids. Scanning electron microscopy images show gypsum particles practically engulfed by hyphal networks, with secondary minerals like strengite (an iron phosphate) forming at the interface.
This matters because conventional chemical extraction methods face several problems. Strong acids dissolve everything indiscriminately—not just phosphorus, but also calcium, iron, silicon and various other elements. The dissolved calcium tends to recombine with phosphate, reducing extraction efficiency. Iron also binds strongly to phosphate, forming compounds that resist further processing. And silicon creates silica gels that physically block access to remaining phosphogypsum particles. All of which helps explain why even aggressive chemical treatments typically leave about 1 per cent residual phosphorus behind.
Biological extraction, by contrast, is selective. A. niger appears to target phosphorus specifically, driven by its nutritional requirements. The fungus needs phosphorus to build cell membranes and synthesize energy-carrying molecules, so it keeps secreting the organic acids necessary to liberate more—a self-sustaining feedback loop. The acidity of phosphogypsum actually helps, creating favourable initial conditions that support vigorous fungal growth.
The biomass itself has potential value. Once the fungus has done its work, the phosphorus-enriched fungal cells could be applied as a slow-release fertiliser or processed for use in biochemical industries. This is rather different from phosphorus dissolved in solution, which requires further concentration and purification steps.
“Recovering phosphorus from industrial waste aligns well with the goals of sustainable agriculture and circular resource use,” says Li. “This approach could reduce environmental risks while supplementing existing phosphorus supplies.”
The experiments were conducted at laboratory scale, admittedly, and several practical questions remain. How would the process perform with phosphogypsum from different sources, given that composition varies depending on the original phosphate rock and processing conditions? What’s the optimal incubation time that balances extraction efficiency against operational costs? Could the technique be scaled up to industrial volumes?
Even so, the findings suggest a biological route for tackling a problem that has vexed the fertiliser industry for decades. The global phosphate fertiliser market generates these mountains of phosphogypsum as an almost inevitable consequence of production, and the waste keeps accumulating with nowhere to go. If filamentous fungi can recover substantial amounts of the trapped phosphorus through relatively simple fermentation processes, that begins to look like a solution rather than just an interesting laboratory curiosity.
There’s a certain symmetry to using biology to extract nutrients from waste generated by the fertiliser industry. Phosphate fertilisers are mined, processed with sulphuric acid, applied to fields—and then much of that phosphorus either binds to soil particles or washes into waterways, where it contributes to algal blooms and dead zones. Perhaps 15 to 30 per cent of applied phosphorus actually reaches crop plants. The rest is effectively lost from agricultural systems, even as the finite reserves of high-grade phosphate rock dwindle.
Closing this loop by recovering phosphorus from industrial waste streams could extend the functional lifetime of existing phosphate reserves. It won’t solve the whole problem, but it might buy time while more fundamental changes in agricultural practices take hold. And if the process also helps remediate the environmental hazards posed by billions of tonnes of stockpiled phosphogypsum, so much the better.
The researchers are now exploring how to optimise extraction efficiency and investigating whether similar approaches might work for other industrial wastes containing valuable but difficult-to-access nutrients. For now, A. niger is demonstrating that what looked like an intractable waste problem might contain the seeds of its own solution—you just need the right organism to unlock it.
Study link: https://www.maxapress.com/article/doi/10.48130/ebp-0025-0018
Discover more from SciChi
Subscribe to get the latest posts sent to your email.