Landmark experiment reveals the precious gem’s potential in computing
For the first time, physicists have demonstrated that information can flow through a diamond wire.
In the experiment, electrons did not flow through diamond as they do in traditional electronics; rather, they stayed in place and passed along a magnetic effect called “spin” to each other down the wire—like a row of sports spectators doing “the wave.”
Spin could one day be used to transmit data in computer circuits—and this new experiment, done at The Ohio State University, revealed that diamond transmits spin better than most metals in which researchers have previously observed the effect.
Researchers worldwide are working to develop so-called “spintronics,” which could make computers simultaneously faster and more powerful.
Diamond has a lot going for it when it comes to spintronics, said lead investigator Chris Hammel, Ohio Eminent Scholar in Experimental Physics at Ohio State. It’s hard, transparent, electrically insulating, impervious to environmental contamination, resistant to acids, and doesn’t hold heat as semiconductors do.
“Basically, it’s inert. You can’t do anything to it. To a scientist, diamonds are kind of boring, unless you’re getting engaged,” Hammel said. “But it’s interesting to think about how diamond would work in a computer.”
The price tag for the diamond wire didn’t reach engagement ring proportions, Hammel confirmed. It cost a mere $100, since it was made of synthetic, rather than natural, diamond.
The findings here represent the first very small step along a very long road that could one day lead to diamond transistors.
But beyond that, this discovery could change the way researchers study spin, Hammel said.
The finding appears in the March 23 issue of the journal Nature Nanotechnology.
Electrons attain different spin states according to the direction in which they’re spinning—up or down. Hammel’s team placed a tiny diamond wire in a magnetic resonance force microscope and detected that the spin states inside the wire varied according to a pattern.
“If this wire were part of a computer, it would transfer information. There’s no question that you’d be able to tell at the far end of the wire what the spin state of the original particle was at the beginning,” he said.
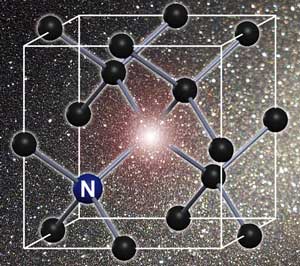
Normally, diamond couldn’t carry spin at all, because its carbon atoms are locked together, with each electron firmly attached to a neighboring electron. The researchers had to seed the wire with nitrogen atoms in order for there to be unpaired electrons that could spin. The wire contained just one nitrogen atom for every three million diamond atoms, but that was enough to enable the wire to carry spin.
The experiment worked because the Ohio State physicists were able to observe electron spin on a smaller scale than ever before. They focused the magnetic field in their microscope on individual portions of the wire, and found that they could detect when spin passed through those portions.
The wire measured only four micrometers long and 200 nanometers wide. In order to see inside it, they set the magnetic coil in the microscope to switch on and off over tiny fractions of a second, generating pulses that created 15-nanometer (about 50-atoms) wide snapshots of electron behavior. They knew that spin was flowing through the diamond when a magnet on a delicate cantilever moved minute amounts as it was alternatively attracted or repelled by the atoms in the wire, depending on their spin states.
Even more surprising was that the spin states lasted twice as long near the end of the wire than in the middle. Based on ordinary experiments, the physicists would expect spin states to last for the same length of time, regardless of where the measurement was made. In this case, spin states inside the wire lasted for about 15 milliseconds, and near the end they lasted for 30 milliseconds.
Hammel’s team suspects that they were able to witness this new effect in part because of how closely they were able to zoom in on the wire. As they focused their tiny window of observation on the tip of the wire, they witnessed spin flowing in the only direction it could flow: into the wire. When they panned along the wire to observe the middle, the “window” emptied of spin twice as fast, because the spin states could flow in both directions—into and out of the wire.
“It’s a dramatically huge effect that we were not anticipating,” Hammel said.
The discovery challenges the way researchers have studied spin for the last 70 years, Hammel explained.
“The fact that spins can move like this means that the conventional way that the world measures spin dynamics on the macroscopic level has to be reconsidered—it’s actually not valid,” he added.
Conventional experiments don’t have the fine resolution to look inside objects as small as the wire used in this study, and so can only look at such objects as a whole. Under those circumstances, researchers can only detect the average spin state: how many electrons in the sample are pointing up, and how many are pointing down. Researchers wouldn’t know the difference if a few electrons in one part of the sample flipped from down to up, and another part flipped from up to down, because the average number of spins would remain the same.
“It’s not the average we want,” Hammel said. “We want to know how much the spins vary, and what is the lifetime of any particular spin state.”
It’s the difference between knowing that an average of one quarter of all spectators in a stadium are standing at any one time, and knowing that individual people are standing and sitting in a pattern timed to form “the wave.”
Nobody could see the spins in diamond before, but this experiment proved that diamond can transport spin in an organized way, preserving spin state—and, thus, preserving information.
The physicists had to chill the wire to 4.2 Kelvin (about -452 degrees Fahrenheit or -269 degrees Celsius) to slow down the spins and to quiet their sensitive detector enough to make these few spins detectable. Many advances would have to be made before the effect could be exploited at room temperature.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.

It is thought to be believed that solid-state computers as opposed to earlier gas- and liquid-state systems; could become major in the future because they can easily be made into different sizes. Currently computers are very small in size; this cannot however, compete with the speed of larger, old-style computers.
The question now that has been asked is whether a “diamond can be a computer’s best friend”? I think the answer is yes!
The diamond computer system featured has two quantum bits (qubits) made of subatomic particles. The traditional computer can encode either a one or a zero; qubits can encode both a one and a zero at the same time. This characteristic allows for the travelling through ‘energy barriers’ and will someday allow computers to perform calculations much faster than old-style computers.
Like all diamonds, the diamond used by the researchers had impurities. These impurities refer to the diamond appearing cloudy and this is a result of substances other than carbon. However, the scientists who have been working on the diamond computer managed to utilize these impurities. This of course adds to the effectiveness of this future proposal.