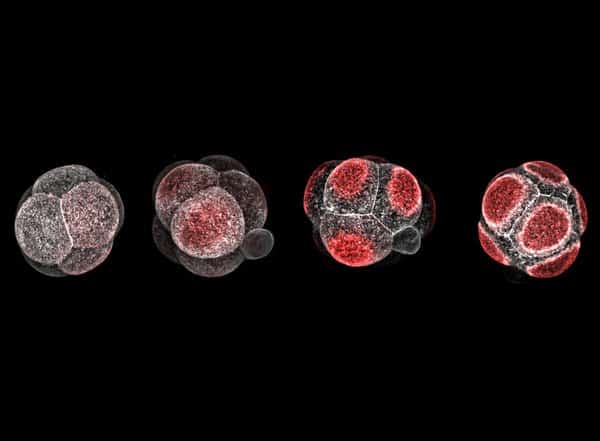
It is estimated that the majority of pregnancies that fail do so within the first seven days after fertilization, before the embryo implants into the uterus. In this time period, a complicated cascade of events occurs with precise timing. One particularly important process is called polarization, when the individual cells that make up the embryo become asymmetrical. Polarization occurs at 2.5 days for mouse embryos and 3.5 days for human embryos.
Just as musicians playing together in an orchestra need to play at the right time—not early, not late—the timing of polarization is critical for proper embryonic function. Studies have shown that if polarization occurs too early or too late, the embryo is less likely to develop properly. Embryos obviously cannot look at a clock to know when it is time to polarize, so how do they “know” when it is time?
A new study from the laboratory of Caltech developmental biologist Magdalena Zernicka-Goetz, Bren Professor of Biology and Biological Engineering, has uncovered the signals that mouse embryos follow to initiate polarization. Understanding the molecular mechanisms underlying embryonic development is critical for understanding how life begins.
“Our lab wants to understand the molecular and cellular mechanisms of the first two weeks of our lives that are critical for the success of our development,” says Zernicka-Goetz. “This is the time that the future body is defined. Our new findings identify the mechanism underlying the very first choice that the embryo’s cells have to take in development.”
A paper describing the research appeared in the journal Science on December 11, 2020.
The work, led by University of Cambridge postdoctoral scholar Meng Zhu, identified two important steps that are necessary for the correct timing of polarization in the mouse embryo.
The first is the zygotic genome activation, or ZGA, which represents the initial “awakening” of the embryonic DNA after it has combined from sperm and egg, with certain genes for development flipped on like a dormant computer booting up. A flood of molecular activity follows ZGA, and during that period, the team found, three specific factors—protein-based structures called Tfap2c, Tead4, and RhoA—work together to initiate polarization.
This research is the first to identify the necessary and sufficient conditions that drive cell polarization. “This has been unknown for a long time,” says Zhu. “Our work illustrates the important molecular basis about the mechanism behind cell polarization and paves the way for answering even more questions about embryonic development, such as what triggers the expression of the three factors we identified? What triggers ZGA, which is upstream of everything?”
Once the team had identified the three factors that initiate polarization, they turned their focus to the polarization process itself. Led by the paper’s second author, Jake Cornwall-Scoones, who was an undergraduate student in the Zernicka-Goetz laboratory at Cambridge before the lab’s transition to Caltech in 2019, the team aimed to develop a quantitative model of the polarization process. Working together with Caltech’s Matt Thomson, assistant professor of biology and biological engineering, Cornwall-Scoones developed a quantitative model of how proteins move throughout the embryo to make it polarized. The model took into account the three factors identified by Zhu and revealed how changes in the levels of these factors then altered movement of the polarization proteins. The model correctly matched microscope imaging of embryos conducted by Zhu.
“It has been so exciting to see Jake’s polarity model come together with the breathtaking quantitative data from Meng and the model system developed by Magda’s group,” says Thomson, who is also a Heritage Medical Research Institute Investigator. “Meng and Jake were able to fit parameters in the model to data and make quantitative predictions about the dynamics of the process and failure modes that can occur in different mutants. This paper is one of the first cases that I am aware of where it has been possible to achieve a truly quantitative understanding of a critical developmental process in the early embryo.”
Zernicka-Goetz’s team is now studying to what extent the polarization timing mechanism in the mouse embryo is similar to the analogous process in human embryos.
The paper is titled “Developmental clock and mechanism of de novo polarization of the mouse embryo.” In addition to Zhu, Cornwall-Scoones, Thomson, and Zernicka-Goetz, additional coauthors are Peizhe Wang and Jie Na of Tsinghua University in China, and Charlotte Handford of the University of Cambridge. Funding was provided by the Wellcome Trust, the European Research Council, the Leverhulme Trust, Open Philanthropy, the Weston Havens Foundation, the National Institutes of Health, the David and Lucile Packard Foundation, the Heritage Medical Research Institute, and the National Key R&D Program of China. Magdalena Zernicka-Goetz is an affiliated faculty member with the Tianqiao and Chrissy Chen Institute for Neuroscience at Caltech.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.