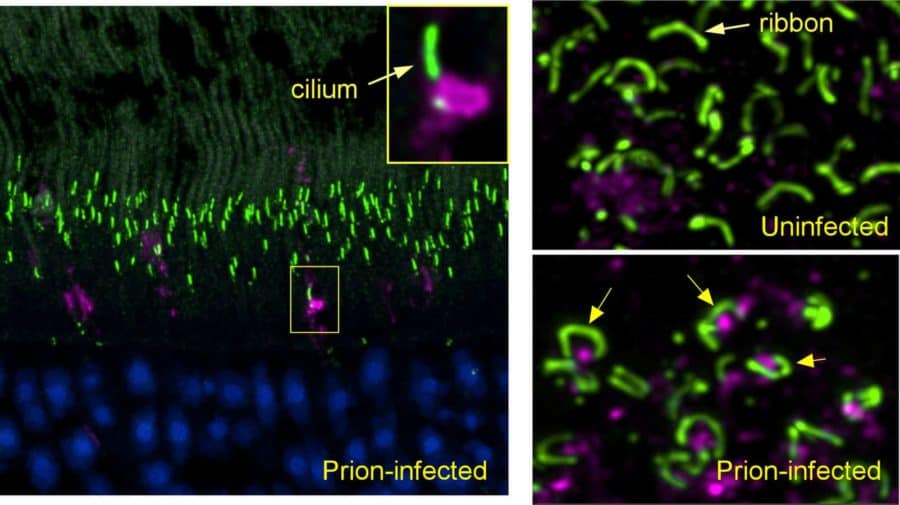
The earliest eye damage from prion disease takes place in the cone photoreceptor cells, specifically in the cilia and the ribbon synapses, according to a new study of prion protein accumulation in the eye by National Institutes of Health scientists. Prion diseases originate when normally harmless prion protein molecules become abnormal and gather in clusters and filaments in the human body and brain.
Understanding how prion diseases develop, particularly in the eye because of its diagnostic accessibility to clinicians, can help scientists identify ways to slow the spread of prion diseases. The scientists say their findings, published in the journal Acta Neuropathologica Communications, may help inform research on human retinitis pigmentosa, an inherited disease with similar photoreceptor degeneration leading to blindness.
Prion diseases are slow, degenerative and usually fatal diseases of the central nervous system that occur in people and some other mammals. Prion diseases primarily involve the brain, but also can affect the eyes and other organs. Within the eye, the main cells infected by prions are the light-detecting photoreceptors known as cones and rods, both located in the retina.
In their study, the scientists, from NIH’s National Institute of Allergy and Infectious Diseases at Rocky Mountain Laboratories in Hamilton, Montana, used laboratory mice infected with scrapie, a prion disease common to sheep and goats. Scrapie is closely related to human prion diseases, such as variant, familial and sporadic Creutzfeldt-Jakob disease (CJD). The most common form, sporadic CJD, affects an estimated one in one million people annually worldwide. Other prion diseases include chronic wasting disease in deer, elk and moose, and bovine spongiform encephalopathy in cattle.
Using confocal microscopy that can identify prion protein and various retinal proteins at the same time, the scientists found the earliest deposits of aggregated prion protein in cone photoreceptors next to the cilia, tube-like structures required for transporting molecules between cellular compartments. Their work suggests that by interfering with transport through cilia, these aggregates may provide an important early mechanism by which prion infection selectively destroys photoreceptors. At a later study timepoint, they observed similar findings in rods.
Prion protein also was deposited in cones and rods adjacent to ribbon synapses just before the destruction of these structures and death of photoreceptors. Ribbon synapses are specialized neuron connections found in ocular and auditory neural pathways, and their health is critical to the function of retinal photoreceptors in the eye, as well as hair cells in the ear.
The researchers say such detailed identification of disease-associated prion protein, and the correlation with retinal damage, has not been seen previously and is likely to occur in all prion-susceptible species, including people.
Next the researchers are hoping to study whether similar findings occur in retinas of people with other degenerative diseases characterized by misfolded host proteins, such as Alzheimer’s and Parkinson’s diseases.
Article
J. Striebel et al. Prion-induced photoreceptor degeneration begins with misfolded prion protein accumulation in cones at two distinct sites: cilia and ribbon synapses. Acta Neuropathologica Communications DOI: 10.1186/s40478-021-01120-x (2021).
Related
J Striebel et al. Microglia are not required for prion-induced retinal photoreceptor degeneration. Acta Neuropathologica Communications DOI: 10.1186/s40478-019-0702-x (2019).
J Carroll et al. Microglia are critical in host defense against prion disease. Journal of Virology DOI: 10.1128/JVI.00549-18 (2018).
Who
Bruce Chesebro, M.D., chief of NIAID’s Laboratory of Persistent Viral Diseases, is available to comment on this study.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.