Massages feel good, but do they actually speed muscle recovery? Turns out, they do. Scientists at the Wyss Institute and Harvard John A. Paulson School of Engineering and Applied Sciences applied precise, repeated forces to injured mouse leg muscles and found that they recovered stronger and faster than untreated muscles, likely because the compression squeezed inflammation-causing cells out of the muscle tissue.
Using a custom-designed robotic system to deliver consistent and tunable compressive forces to mice’s leg muscles, researchers at Harvard’s Wyss Institute for Biologically Inspired Engineering and SEAS found that this mechanical loading (ML) rapidly clears immune cells called neutrophils out of severely injured muscle tissue. This process also removed inflammatory cytokines released by neutrophils from the muscles, enhancing the process of muscle fiber regeneration. The research is published in Science Translational Medicine.
“Lots of people have been trying to study the beneficial effects of massage and other mechanotherapies on the body, but up to this point it hadn’t been done in a systematic, reproducible way,” said first author Bo Ri Seo, who is a postdoctoral fellow in the lab of Dave Mooney at the Wyss Institute and SEAS. “Our work shows a very clear connection between mechanical stimulation and immune function. This has promise for regenerating a wide variety of tissues including bone, tendon, hair, and skin, and can also be used in patients with diseases that prevent the use of drug-based interventions.”
A more meticulous massage gun
Seo and her co-authors started exploring the effects of mechanotherapy on injured tissues in mice several years ago, and found that it doubled the rate of muscle regeneration and reduced tissue scarring over the course of two weeks. Excited by the idea that mechanical stimulation alone can foster regeneration and enhance muscle function, the team decided to probe more deeply into exactly how that process worked in the body, and to figure out what parameters would maximize healing.
They teamed up with soft robotics experts in the Harvard Biodesign Lab, led by Wyss Associate Faculty member Conor Walsh to create a small device that used sensors and actuators to monitor and control the force applied to the limb of a mouse. The team experimented with applying force to mice’s leg muscles via a soft silicone tip and used ultrasound to get a look at what happened to the tissue in response. They observed that the muscles experienced a strain of between 10 to 40 percent, confirming that the tissues were experiencing mechanical force. They also used those ultrasound imaging data to develop and validate a computational model that could predict the amount of tissue strain under different loading forces.
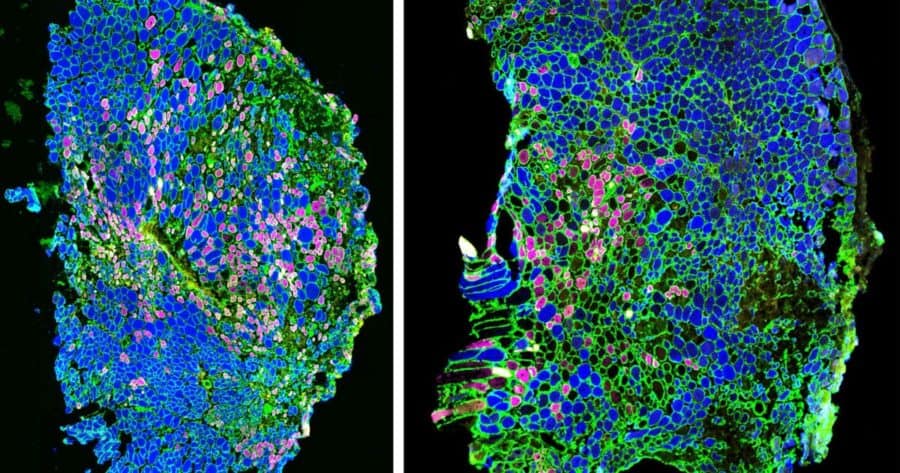
They then applied consistent, repeated force to injured muscles for 14 days. While both treated and untreated muscles displayed a reduction in the amount of damaged muscle fibers, the reduction was more pronounced and the cross-sectional area of the fibers was larger in the treated muscle, indicating that treatment had led to greater repair and strength recovery. The greater the force applied during treatment, the stronger the injured muscles became, confirming that mechanotherapy improves muscle recovery after injury. But how?
Evicting neutrophils to enhance regeneration
To answer that question, the scientists performed a detailed biological assessment, analyzing a wide range of inflammation-related factors called cytokines and chemokines in untreated vs. treated muscles. A subset of cytokines was dramatically lower in treated muscles after three days of mechanotherapy, and these cytokines are associated with the movement of immune cells called neutrophils, which play many roles in the inflammation process. Treated muscles also had fewer neutrophils in their tissue than untreated muscles, suggesting that the reduction in cytokines that attract them had caused the decrease in neutrophil infiltration.
The team had a hunch that the force applied to the muscle by the mechanotherapy effectively squeezed the neutrophils and cytokines out of the injured tissue. They confirmed this theory by injecting fluorescent molecules into the muscles and observing that the movement of the molecules was more significant with force application, supporting the idea that it helped to flush out the muscle tissue.
To pick apart what effect the neutrophils and their associated cytokines have on regenerating muscle fibers, the scientists performed in vitro studies in which they grew muscle progenitor cells (MPCs) in a medium in which neutrophils had previously been grown. They found that the number of MPCs increased, but the rate at which they differentiated (developed into other cell types) decreased, suggesting that neutrophil-secreted factors stimulate the growth of muscle cells, but the prolonged presence of those factors impairs the production of new muscle fibers.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.