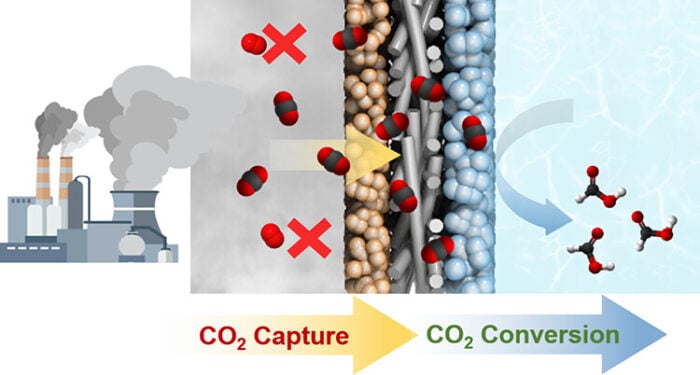
Somewhere right now, a factory stack is belching smoke that looks no different from any other day. But if researchers at Korea’s KENTECH have their way, that plume (along with the carbon dioxide it carries) will soon become the raw material for tomorrow’s chemicals and fuels. The obstacle hasn’t been the capturing part. It’s been what comes next.
“Capturing CO2 from the air should be relatively simple, after all, plants do it all the time,” the team notes. “But converting the gas into something useful is difficult, and it is a crucial step in ensuring that carbon capture methods are widely implemented.” For decades, this catch-22 has stalled the whole enterprise. Capture without conversion is just carbon lockup. Conversion without capture means starting with pure, expensive CO2.
The research published this week in ACS Energy Letters breaks that deadlock with something deceptively simple: a layered electrode that captures and converts CO2 simultaneously, doing both jobs in the same place without requiring the gas to be separated, concentrated, or purified first. The team, led by Myoung Hwan Oh, Wonyong Choi, and colleagues at the Korea Institute of Energy Technology, designed this composite electrode with three distinct layers, each doing specific work. A specialized carbon-capturing material sits on the gas-facing side, followed by gas-permeable carbon paper, then a catalytic tin oxide layer that performs the actual chemical conversion.
The simplicity is the point. “This work shows that carbon capture and conversion do not need to be treated as separate steps,” Choi explains. “By integrating both functions into a single electrode, we demonstrate a simpler pathway for CO2 utilization under realistic gas conditions.” Those realistic conditions matter enormously. Most existing conversion technologies demand highly concentrated CO2. Industrial waste streams diluted with nitrogen and oxygen simply won’t cooperate with them.
In laboratory tests under pure, concentrated CO2, the new electrode proved about 40 percent more efficient than current alternatives, converting CO2 directly into formic acid at rates of 250 micromoles per hour per square centimeter, with an efficiency of 98 percent. That’s respectable. But the real test came when researchers fed it simulated flue gas: the actual mixture belching from industrial stacks (15 percent CO2, 8 percent oxygen, 77 percent nitrogen).
The results split the field. Other electrodes produced essentially nothing. The new design kept producing formic acid at meaningful rates. The oxygen shouldn’t work. It poisons the reaction, spinning off toward unwanted side reactions instead of letting the CO2 through to the catalyst layer. Yet the oxygen-functionalized carbon in the capture layer acts as a selective barrier, letting CO2 molecules preferentially drift through while blocking oxygen. A bit like having a bouncer that only recognizes certain molecules. Computer simulations revealed the mechanism: the carbon’s chemical structure creates favorable electrostatic interactions that attract CO2 molecules while repelling oxygen, a nanoscale discrimination that transforms the whole efficiency calculation.
Even more striking, the electrode worked with ultradilute CO2 (down to 1 percent concentration and even at 400 parts per million, roughly the level in ambient air). This opens an entirely new possibility: direct-air capture without the expensive separation step. Donglai Pan, Jaeyeon Yang, and their colleagues tested this not just in the lab but through sustained operation, confirming that production remained stable whether the gas was concentrated industrial exhaust or the thin CO2 everywhere around us.
The target product, formic acid, isn’t glamorous but it’s useful. It’s a chemical workhorse in fuel cells, pharmaceuticals, dyes, and countless industrial syntheses. By turning concentrated waste into a distributed feedstock, the technology could make carbon capture economically sensible. Current approaches require expensive infrastructure to isolate pure CO2, then compress and transport it. With this electrode, factories could use flue gas directly. The math shifts dramatically once conversion exceeds about 25 percent efficiency. Below that, separate capture and conversion makes sense. Above it, integrated systems win. This electrode operates above that threshold.
The researchers think the approach generalizes. They’ve already tested it with different catalyst materials and different products, showing the adsorption layer principle isn’t locked to formic acid. The same logic could work for capturing nitrogen oxides, converting oxygen to hydrogen peroxide, or oxidizing methane into methanol and formic acid. That means turning industrial and agricultural waste into chemical feedstocks. Each application would require engineering the selectivity differently, tuning which molecules the capture layer favors, but the fundamental insight carries: integrate capture and conversion, let chemistry do the work of separation, make the system work with dilute, realistic gas mixtures.
That’s the real promise here. Not a breakthrough in abstract efficiency, but a solution to why carbon capture remains stuck on shelves rather than running in factories. Climate solutions don’t advance because scientists achieve perfect laboratory performance. They advance when someone figures out how to make them work under the messy conditions of the real world, where gases come in mixtures, concentrations are low, and every step of purification burns money and energy. This electrode moves that needle. It won’t solve climate change alone. Nothing could. But it removes one significant barrier from a problem that has too many of them already.
Study: https://doi.org/10.1021/acsenergylett.5c03504
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.