Anna Caroline Aguiar leans over blood samples fresh from infected patients in Rondônia, Brazil’s malaria heartland. Under her microscope, something remarkable is happening. The parasites that normally twist and develop inside mosquito guts are simply stopping. “We’ve been studying this compound for five years,” she says, and every time we test it, it surprises us.
What Aguiar and her team at the Federal University of São Paulo have found is rare in malaria research: a single molecule that attacks the parasite at all three critical stages of its life cycle. It kills Plasmodium in human livers before symptoms appear, wipes it from blood during infection, and stops transmission to mosquitoes. That triple action could reshape how we fight a disease that still kills 600,000 people a year.
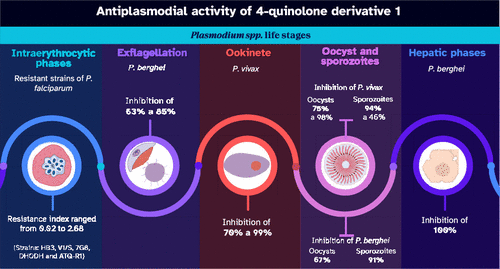
The compound comes from an unlikely source. Natural 4-quinolones appear in certain plants and microorganisms, and researchers have been tinkering with their structure for decades. Aguiar’s team modified a 2-substituted-4-quinolone by adding an eleven-carbon chain, creating what they now call compound 1. The change made all the difference. When they tested it against Plasmodium vivax (the dominant malaria species in Brazil that can’t be grown continuously in labs) the molecule showed an inhibition rate of 97 per cent against early parasite forms.
Here’s why that matters. Plasmodium has evolved into one of nature’s most cunning parasites. When an infected mosquito bites you, it injects sporozoites that race to your liver. There they multiply silently for days. Then they burst back into your bloodstream, invading red blood cells and causing the fever, chills and potentially deadly complications we call malaria. Some parasites transform again into gametocytes, sexual forms that wait in your blood for the next mosquito bite. Inside that mosquito, they reproduce and become sporozoites again, ready to infect another person.
Most malaria drugs hit just one stage of this cycle. Compound 1 disrupts all three. In laboratory tests on human liver cells infected with Plasmodium berghei, the molecule prevented infection at nanomolar concentrations of around 250 billionths of a gram per litre. When Aguiar’s team tested it on blood from six patients infected with P. vivax, then fed that blood to laboratory-reared mosquitoes through artificial membranes, the compound reduced oocyst formation in mosquito midguts by 95 per cent at higher doses.
The molecule works by sabotaging the parasite’s powerhouse. It targets an enzyme complex called cytochrome bc1 in the parasite’s mitochondria, blocking the production of pyrimidines, the building blocks of DNA. Without DNA synthesis, the parasite can’t replicate. Another important aspect is that this molecule is highly selective, says Aguiar, adding that it acts on the parasite’s mitochondria but not on those of humans.
That selectivity shows up in toxicity tests. Rafael Guido at the University of São Paulo points out that compound 1 has a selectivity index above 120, meaning it’s more than 120 times more toxic to parasites than to human cells. When the researchers tested it against multiple drug-resistant Plasmodium falciparum strains, including ones resistant to the current cytochrome bc1 inhibitor atovaquone, the compound maintained submicromolar activity.
Transmission blocking is where compound 1 truly shines. Direct membrane feeding assays, where laboratory mosquitoes feed on drug-treated blood from infected patients, showed the compound prevented parasite development inside the insect vector. Even if an infected person treated with compound 1 is bitten by a mosquito, the parasite can’t complete its life cycle to infect someone else. In tests with mice infected with P. berghei, mosquitoes that fed on compound 1-treated animals showed a 91 per cent reduction in salivary gland infection — the final stage before transmission.
Aguiar emphasizes the rarity of this profile. Malaria patients typically need different medications for different stages. Treatment drugs clear blood infections but don’t prevent relapses from dormant liver parasites. Prophylactic drugs protect the liver but don’t cure active infections. And most drugs do nothing to stop transmission. This compound combines treatment and transmission-blocking potential, she says, with possible use for prevention.
The molecule still faces a long development path. It needs optimization for better absorption and stability in the body, safety testing in larger animals, and eventually human clinical trials. Drug development from promising compound to approved medicine typically takes a decade and costs hundreds of millions. But the evidence so far suggests compound 1 and its chemical relatives deserve that investment.
Rafael Guido is cautiously optimistic. The molecule is an excellent candidate, and the evidence warrants investment in the future development of a drug, he says. This is because, although there’s treatment for the disease, it’s a very well-adapted parasite capable of developing resistance to existing drugs. As resistance spreads to current antimalarials, molecules that attack multiple life stages become not just useful but essential. Finding one that does it this well is the kind of discovery that reminds you why researchers keep looking.
Study link: https://pubs.acs.org/doi/10.1021/acsomega.5c08663
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.