The mitral valve sits between the two chambers on the left side of your heart, a pair of leaflets that open and snap shut roughly seventy times a minute for your entire life. When those leaflets stop closing properly, blood flows the wrong way, pooling and straining a system built for one-directional pressure. Fixing it requires threading instruments through a catheter while the heart keeps beating, navigating a space full of fine tendinous strings that tether the valve to the chamber wall. Getting it wrong in a patient is not an option.
Learning to get it right, though, has always required practice on something. That something is usually an animal organ, a cadaver, or a computer screen. None of them is ideal: animal hearts aren’t your patient’s heart, cadavers don’t beat, and computer screens don’t give you the physical resistance of tissue that pushes back.
A team at Washington State University has built an alternative. Their model is a 3D-printed replica of the left side of the heart, made from a silicone-based ink tuned to approximate the mechanical properties of cardiac muscle. It has a left atrium, a left ventricle and a mitral valve, all in roughly the right proportions and geometry, all soft enough to handle the way a real heart would. Small pneumatic actuators embedded in the ventricular walls inflate and contract in sequence, squeezing the chambers and driving the mitral valve open and shut. The whole thing runs at 70 beats per minute. “It’s very useful for doctors and surgeons to practice when the heart is still beating, especially for minimally invasive surgery,” says Kaiyan Qiu, Berry Family Assistant Professor in WSU’s School of Mechanical and Materials Engineering and the paper’s corresponding author.
The problem with earlier synthetic models isn’t that they don’t beat. Some of them do. But they’ve generally been built using molds, which limits what shapes are actually achievable. “There have been other, synthetic models that are mostly mold-casted, and one of the main limitations there is that they cannot do some of the more complex curvatures that you see in the heart,” says Alejandro Guillen Obando, a PhD candidate in the same department and the paper’s first author.
The curvatures matter. The left ventricle isn’t a tube or a box; it bends and tapers in ways that affect how instruments behave inside it. The mitral valve annulus is asymmetric. The chordae tendineae, those tendinous strings, branch off the valve leaflets at irregular angles and create exactly the kind of obstruction that trips up surgical instruments during minimally invasive procedures. Getting any of that wrong in training produces habits that don’t transfer to the operating room. “Our layer-by-layer approach in 3D printing allows us to add more curvature and make the chambers simulate a real heart,” says Guillen Obando.
To make the model work, the WSU team had to solve several problems at once. The ink needed to be soft enough to behave like heart tissue (Young’s modulus of around 123 kilopascals, close to the myocardium in its contracting phase) but stiff enough not to collapse during printing. They settled on a 1:1 mixture of silicone sealant and silicone grease, printed layer by layer using a technique called direct ink writing. The mitral valve was printed separately with a deliberate defect in one of its leaflets, then sutured into the ventricle; the sutures themselves thread through the wall of the chamber and act as the chordae tendineae. Once those were in place, the team resumed printing, building the atrium on top. The McKibben actuators, cylindrical pneumatic devices that contract axially when pressurised, were embedded in channels within the ventricular wall before the atrium was added.
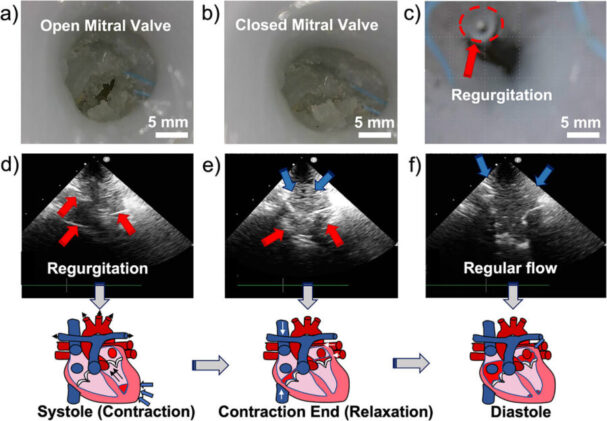
The model also has customised flexible pressure sensors built into the ventricle, water-resistant and laser-cut for precision, which track pressure changes through the cardiac cycle. When the team tested the repair procedure, inserting a clip-like device into the defective mitral valve, the sensors registered an increase in systolic pressure from 45.2 to 67.4 millimetres of mercury, and ultrasound imaging showed that the backward flow of imitation blood had been substantially reduced. Ejection fraction, the proportion of blood the ventricle pushes out with each beat, climbed from 48 per cent to 53 per cent, putting it into the range considered normal in a healthy human heart. “In our case, this model is the first fully synthetic model that, without any assistance of animal models, mimics the complete left side of the heart,” says Qiu. “We were able to incorporate both the anatomic features and the dynamic functions.”
About 800,000 Americans have heart surgery each year. Heart disease remains the leading cause of death in the US.
There are still limits. The model currently can’t generate the high pressures a real left ventricle produces, partly because it lacks the arterial elasticity of the real thing and partly because the purely synthetic system can’t replicate every aspect of cardiac fluid dynamics. The systolic pressures the team recorded are lower than those measured in other studies using animal cardiac tissue to assist the pumping.
What the model can do is provide a reusable, patient-specific platform that doesn’t depend on animal tissue and that a surgeon can actually practice a repair on, with instrumentation, under ultrasound guidance. The team has filed a provisional patent and is now working on a four-chamber version with all four valves; they plan to bring medical professionals into the testing process for procedure-specific rehearsals.
The bigger prospect is that the printing approach lends itself to customisation: scan a particular patient’s heart before a procedure, print a replica with the right geometry, hand it to the surgical team a week before the operation. Whether that becomes practical depends on how much work the four-chamber version requires, and whether the hemodynamic performance can be brought closer to the real thing. For a device that currently just runs at 70 beats per minute on a lab bench, that’s quite a distance to close.
Study link: https://advanced.onlinelibrary.wiley.com/doi/10.1002/admt.70885
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.