Gene therapy just leapt past a barrier that’s held it back for years. Researchers in China have worked out how to pack oversized genes into the viral delivery vehicles that doctors use to treat genetic diseases, a trick that could unlock treatments for hundreds of conditions previously considered untreatable.
The breakthrough centres on adeno-associated viruses, or AAVs. These microscopic packages are the workhorses of gene therapy because they’re safe and effective at ferrying therapeutic DNA into cells. There’s a catch, though. They can only carry small genetic cargoes, typically under 5 kilobases (a measure of DNA length).
That’s fine for some diseases. But roughly 37.5% of genes linked to autism are bigger than that limit. Same goes for genes behind certain epilepsies, muscular dystrophies and hundreds of other inherited conditions. For years, scientists have been trying workarounds – splitting genes into pieces, using multiple viruses, hoping the fragments reassemble properly inside cells. The results have been, to put it mildly, hit and miss.
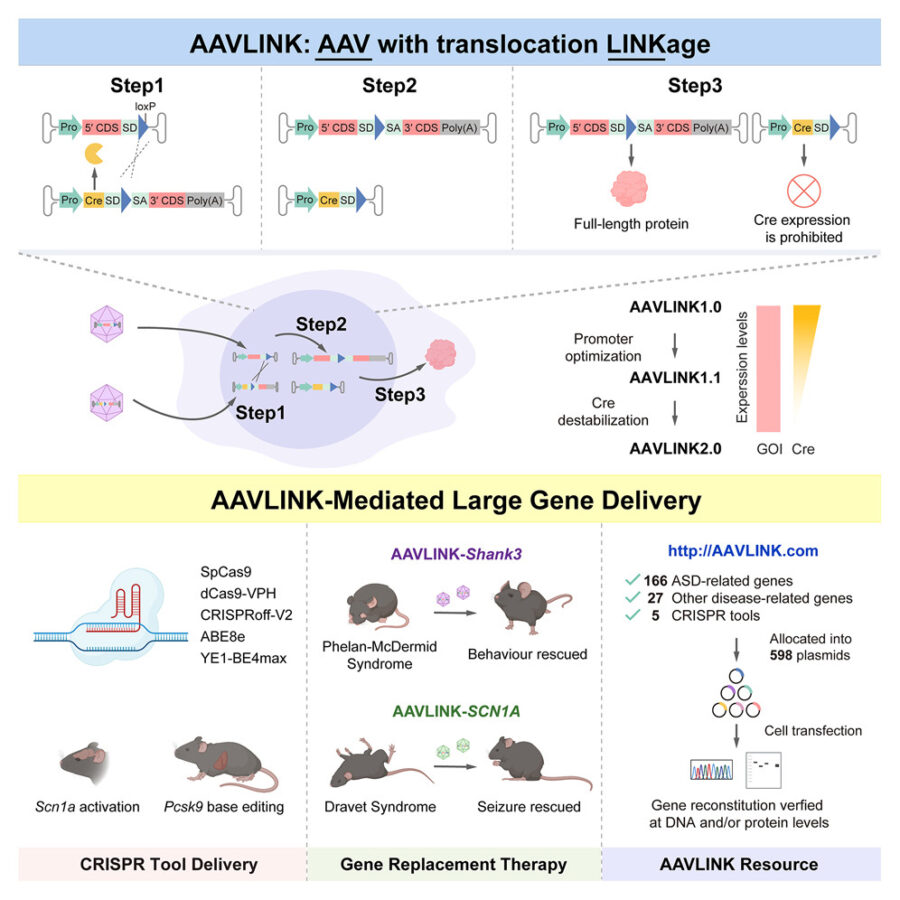
Now Zhonghua Lu at the Shenzhen Institutes of Advanced Technology and his colleagues have developed what they’re calling AAVLINK, which uses a molecular cutting-and-pasting system to stitch together oversized genes with remarkable efficiency. The team split large therapeutic genes into two or three chunks, packaged them into separate AAV particles, then programmed the fragments to recombine inside target cells using a protein called Cre recombinase.
The clever bit is what happens after the fragments join up. The recombination process automatically switches off Cre production, which addresses a longstanding safety concern about these systems. And unlike previous split-gene approaches, AAVLINK produces very few incomplete or malformed proteins, the sort of molecular junk that can cause problems in cells.
To test whether this actually works in living animals, the team focused on two devastating childhood conditions. Phelan-McDermid syndrome is caused by mutations in SHANK3, a gene too large for standard AAV delivery (the syndrome typically involves intellectual disability and autism-like behaviours). When Lu’s group used AAVLINK to deliver intact SHANK3 to mice with the condition, the animals showed fewer repetitive behaviours and better motor coordination. Hardly a cure, perhaps, but meaningful improvements.
The second test was even more dramatic. Dravet syndrome is a severe form of childhood epilepsy, and more than 80% of cases trace back to a faulty copy of SCN1A, another gene that exceeds AAV’s carrying capacity. Mice with Dravet syndrome usually die within three weeks. The team delivered working copies of SCN1A using AAVLINK, and the treated mice lived longer, had fewer seizures and showed more normal brain activity.
What’s particularly useful – if you’re thinking about translating this to human patients – is that the researchers built a whole library of AAVLINK vectors covering 193 disease-associated genes plus five different CRISPR gene-editing tools. They’ve tested them all in cultured cells to verify the genes reassemble properly. That’s quite a resource for anyone wanting to develop new therapies.
The approach also worked in monkeys, which suggests it might translate to humans reasonably well. Lu’s team injected the split-gene system into cynomolgus macaques and saw robust gene expression in the brain, roughly comparable to what they’d seen in mice.
There are competing technologies, of course. A team in the US recently described something called StitchR, which uses a different molecular mechanism to join gene fragments together. But in head-to-head comparisons, AAVLINK produced more complete proteins and fewer unwanted by-products. Whether that advantage holds up in human trials remains to be seen.
The researchers have also developed a second-generation version called AAVLINK2.0, which degrades the Cre protein even more rapidly after it’s done its job. That’s meant to address lingering worries about having this foreign protein hanging about in cells long-term, though it’s worth noting Cre has been used in thousands of biology experiments over the years without causing obvious problems.
Clinical applications are still some way off. The team needs to test AAVLINK in larger animals, work out optimal dosing, and demonstrate long-term safety. Regulatory approval will take years, assuming it comes at all. And there’s always the question of cost; AAV-based gene therapies already run to millions of pounds per patient.
Still, for families dealing with genetic diseases that until now have been out of reach, this sort of technical advance offers something rare in medicine. Genuine hope.
Study link: https://www.cell.com/cell/fulltext/S0092-8674(25)01488-6
Discover more from SciChi
Subscribe to get the latest posts sent to your email.