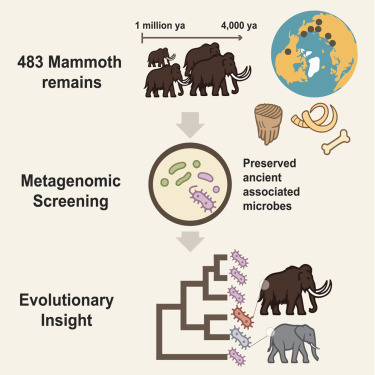
Imagine cradling a fossilized mammoth tooth more than one million years old and learning it still carries microbial traces. That is exactly what an international team led by Stockholm University has discovered, reporting in Cell that woolly and steppe mammoth remains preserve the oldest host-associated bacterial DNA ever recovered.
The findings reveal that microbial lineages survived alongside mammoths for hundreds of thousands of years, and some may even have caused disease.
Unlocking Microbes From The Deep Past
The Centre for Palaeogenetics, a joint initiative of Stockholm University and the Swedish Museum of Natural History, analyzed microbial DNA from 483 mammoth remains, 440 of them sequenced for the first time. Among the collection was a steppe mammoth that lived about 1.1 million years ago. Using genomic and bioinformatic methods, the researchers filtered out post-mortem colonizers to identify microbes that truly lived within the animals.
“Imagine holding a million-year-old mammoth tooth. What if I told you it still carries traces of the ancient microbes that lived together with this mammoth?” said Benjamin Guinet, lead author and postdoctoral fellow at the Centre for Palaeogenetics.
The analyses revealed six microbial clades that persisted across both time and geography, including relatives of Actinobacillus, Pasteurella, Streptococcus, and Erysipelothrix. Some of these are known pathogens in modern elephants. One Pasteurella-related bacterium resembles a microbe that has caused lethal outbreaks among African elephants, raising the possibility that similar infections may have afflicted mammoths.
A Disease Record Written In Bacteria
Perhaps most striking was the reconstruction of partial Erysipelothrix genomes from a 1.1-million-year-old steppe mammoth. This represents the oldest known host-associated microbial DNA ever recovered. Given the fast evolution of bacteria, the recovery of such sequences across a million-year divide pushes the boundaries of paleogenomic research.
“As microbes evolve fast, obtaining reliable DNA data across more than a million years was like following a trail that kept rewriting itself,” said Tom van der Valk, senior author of the study. “Our findings show that ancient remains can preserve biological insights far beyond the host genome.”
While it is difficult to determine exactly how these microbes affected mammoth health, the research points to a long coexistence between host and microbe. This endurance suggests some pathogens shadowed mammoth evolution until the species’ final days on Wrangel Island roughly 4,000 years ago.
A New Window Into Ancient Ecosystems
The study expands what can be learned from ancient DNA. Beyond reconstructing mammoth genomes, scientists can now probe their microbiomes. These microbial companions could reveal much about adaptation, immunity, and even extinction pressures in Pleistocene ecosystems.
“This work opens a new chapter in understanding the biology of extinct species. Not only can we study the genomes of mammoths themselves, but we can now begin to explore the microbial communities that lived inside them,” said Love Dalén, professor of evolutionary genomics at the Centre for Palaeogenetics.
By peeling back this hidden biological layer, researchers gain a richer picture of ancient life: the mammoth not as a lone giant, but as a host carrying entire microbial worlds within it.
Explainer: How Ancient Microbial DNA Survives
DNA is fragile, breaking down after death due to heat, moisture, and microbes. Yet in permafrost and other cold, stable environments, fragments can persist for hundreds of thousands, even millions of years. Ancient DNA researchers use strict contamination controls, damage pattern checks, and comparative analysis to separate authentic sequences from modern intruders. In this study, partial bacterial genomes were painstakingly reconstructed from mammoth remains, showing that not only host DNA but also the microbial passengers within them can survive the march of deep time.
Journal: Cell. DOI: 10.1016/j.cell.2025.08.003
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.