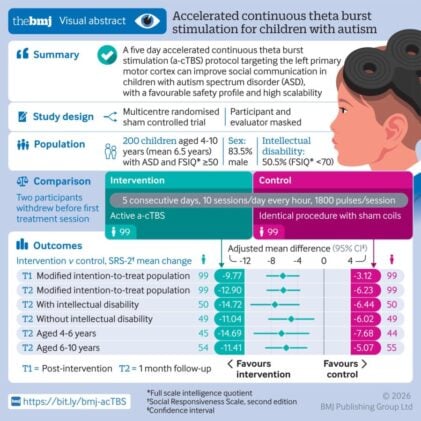
The sessions last two minutes. Just 120 seconds of magnetic pulses directed at the left side of the skull, ten times a day, five days running. The cumulative total comes to 90,000 pulses delivered over a working week. That’s the unremarkable-sounding arithmetic behind a clinical trial published this week in The BMJ that is drawing real attention from the autism research community, not for its elegance but for who it actually worked on: children as young as four, including a third who also had intellectual disability, a population that tends to get quietly excluded from this sort of research.
Social communication impairment sits at the heart of autism spectrum disorder, and it has remained stubbornly resistant to drug treatment. Behavioural therapy works, but only inconsistently, and it works best in children who are higher functioning, who start young, and whose families can afford the intensity of it. One in three autistic people also has intellectual disability. For them, the options narrow considerably.
A New Target in the Motor Cortex
The technique being tested is called accelerated continuous theta burst stimulation, or a-cTBS, a variant of repetitive transcranial magnetic stimulation that has already found a foothold in depression treatment. The team at Xinhua Hospital in Shanghai, led by professor Fei Li, chose an unusual brain target: the left primary motor cortex, the region most associated with movement. That choice needs some explaining. The motor cortex, it turns out, does considerably more than move muscles. It connects to regions involved in language processing, social cognition, and emotion, including the amygdala and orbitofrontal cortex. Motor cortex abnormalities are also consistently observed in autism. More practically, this region can be precisely located using a simple procedure called motor evoked potential detection, without any of the expensive neuronavigation equipment that makes other brain stimulation approaches impossible to scale.
Two hundred children across three Chinese hospitals were enrolled, roughly half with IQs below 70. They were randomised to receive either real stimulation or sham treatment (coils modified to produce the same sound and sensation without the magnetic field). Neither the children, their caregivers, nor the evaluators knew which group was which.
The results, measured using a standard caregiver-rated scale for autism social symptoms, were clear enough that they survived multiple sensitivity analyses. Children receiving active stimulation showed significantly greater reductions in social impairment scores, both immediately after the five days and at a one-month follow-up. The average treatment effect exceeded the threshold the researchers estimated as clinically meaningful. Language abilities also improved, at least among the roughly 60 percent of children who had enough expressive language to complete the relevant tests.
Speed as a Design Feature
The acceleration built into the protocol matters more than it might seem. Standard transcranial magnetic stimulation typically requires sessions of 20 minutes or more, sustained positioning, and a treatment course stretching across weeks or months. That combination has historically made it almost impossible to recruit young children, let alone those with intellectual disability, who find staying still for extended periods genuinely difficult. Reducing each session to two minutes changes that calculus entirely. Ninety-six percent of children completed the full course, which is a high adherence rate by any standard in paediatric research.
The research team concluded the protocol represents “a viable and scalable therapeutic option for children with autism spectrum disorder, including those with intellectual disability” and describes it as “a major advancement towards equitable autism care worldwide.”
Those are fairly strong claims for a single trial with a one-month follow-up, and reviewers in a linked editorial were rather more measured. Researchers commenting from Hong Kong acknowledged the findings show promise but were careful to note that “a-cTBS should not replace psychosocial support or educational adaptation.” The technique, they suggested, “may become an important component of a multimodal pathway for children with autism with significant social communication difficulties,” provided the results are “further replicated and integrated thoughtfully with behavioural care.” That’s a meaningful caveat. Effect sizes, while statistically robust, were in the small-to-moderate range for secondary language outcomes. The primary social communication scores showed larger effects, but caregiver-rated measures are notoriously susceptible to expectancy bias, even in sham-controlled designs where masking appeared reasonably effective.
What It Didn’t Change
Notably, the stimulation did nothing for restricted and repetitive behaviours, the other core symptom cluster in autism. This is probably not a surprise given the neural circuitry involved: repetitive behaviours are tied to cortico-striatal-thalamo-cortical loops that have limited connectivity with the motor cortex. The study also found no significant improvement on broader adaptive behaviour scales, which measure things like daily living skills and socialisation in real-world contexts. Whether those harder-to-shift outcomes might improve with longer follow-up or additional sessions remains an open question.
Side effects were more common in the active group than the sham group (about 55 percent versus 29 percent), with restlessness and scalp discomfort accounting for most of them. All resolved on their own. One child experienced an arm spasm during the final day’s treatment; it passed within minutes and didn’t recur. The safety profile, overall, appears acceptable for a paediatric population, though the follow-up period of one month is too short to say much about effects beyond that window.
What’s perhaps most significant about this trial isn’t the effect size, which is promising but not dramatic, nor the mechanism, which remains not fully understood. It’s the fact that it enrolled the children who most need something to work. The typical autism neuroimaging or neuromodulation study recruits adolescents or adults, without intellectual disability, who can hold still for long procedures and follow complex instructions. Young children with intellectual disability rarely appear in these datasets. A treatment that might actually reach them, that doesn’t require six months of weekly clinic visits or specialist neuronavigation equipment, is worth taking seriously. Whether it will hold up in larger, longer, more diverse trials is still to be seen; but as a signal that the motor cortex might be a tractable target for autism’s social deficits, the data is harder to dismiss than most.
Source: Tan et al., The BMJ, 2026. doi:10.1136/bmj-2025-086295
Frequently Asked Questions
Why would stimulating the motor cortex help with social skills in autism?
The motor cortex does more than control movement. It has connections to brain regions involved in language processing, emotion, and social cognition, and abnormal motor cortex activity is a consistent finding in autism research. The researchers also chose this target partly for practical reasons: it can be precisely located without expensive imaging equipment, making the approach far more scalable than previous brain stimulation protocols that required complex neuronavigation systems.
How is this different from earlier attempts to use brain stimulation for autism?
Most previous trials targeted the dorsolateral prefrontal cortex and enrolled adolescents or adults without intellectual disability who could cooperate with lengthy sessions. This protocol specifically designed around children as young as four, including those with intellectual disability, by reducing each stimulation session to just two minutes. That’s a significant engineering change that opens the treatment to a population historically excluded from this research.
Could this replace behavioural therapy for autistic children?
Experts reviewing the trial explicitly cautioned against that interpretation. Brain stimulation of this kind is unlikely to replace speech therapy, educational support, or other established interventions. The more plausible picture, if the results replicate, is that it becomes one component of a broader treatment plan, potentially making children more responsive to behavioural approaches by improving the underlying social communication difficulties those therapies address.
What are the main reasons to be cautious about these results?
The follow-up period was only one month, which is too short to establish whether improvements last. The primary outcome relied on caregiver ratings, which are vulnerable to expectancy effects even in blinded trials. More than 80 percent of participants were boys, limiting what can be said about girls. And the study was conducted across three hospitals in China, so it’s not yet clear how well the results will transfer to different healthcare settings and populations.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.