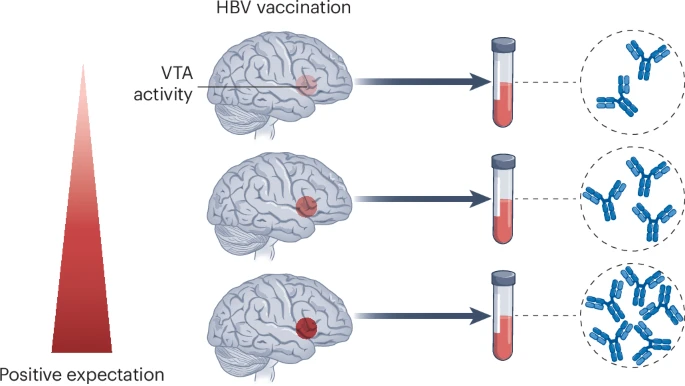
The brain scan showed something unexpected. The volunteer was lying inside the fMRI machine, mentally rehearsing strategies to activate his reward circuitry: imagining a tropical holiday, recalling moments of joy. A tiny region deep in his midbrain lit up like a beacon. Three weeks later, after getting a hepatitis B vaccine, his antibody levels climbed significantly higher than most other participants’. The connection seemed almost absurd: think happy thoughts, build better immunity?
That’s exactly what Nitzan Lubianiker and colleagues at Tel Aviv University demonstrated in a double-blind randomized trial published this month. They studied 85 participants who underwent neurofeedback training to control their brain activity. The people who most effectively ramped up activity in the ventral tegmental area (the VTA, a reward-processing hub no bigger than a cherry pit) showed the strongest immune responses to vaccination. The correlation was specific, measurable, and entirely new in humans.
These findings push decades of placebo research into new territory. Scientists have extensively documented the clinical benefits of positive expectations, but they’ve only begun to understand the underlying neural mechanisms. This study demonstrates for the first time in humans that conscious mental strategies can engage specific reward circuitry to measurably influence immune outcomes.
The Mouse That Fought Infection With Optimism
The human findings didn’t emerge from nowhere. Researchers had already glimpsed this brain-immune link in mice, where the evidence was more direct. (You can manipulate a mouse brain in ways that would be ethically impossible in people.) In 2016, Asya Rolls and colleagues at the Technion in Haifa used chemogenetics to artificially activate dopamine neurons in the mouse VTA. The result? Mice cleared bacterial infections faster and showed reduced tumor growth.
A year later, another study revealed something remarkable. When researchers optogenetically triggered reward-related VTA activity (or even just let male mice encounter females, which naturally activates this circuit), it induced a proinflammatory state. Block VTA activity, and these immune changes vanished. The reward system, it turned out, didn’t just make mice feel good about potential mates. It prepped their bodies to handle the microbial challenges that might follow social encounters.
But rodent brains and human brains are different beasts. Correlation studies in people had hinted at links between reward-related neural activity and immune markers like inflammation, yet nobody had demonstrated causation. Could humans consciously harness their reward circuitry to shape immunity?
Teaching the Brain to Boost Itself
Enter neurofeedback. The technique works like a mental gym. Participants watch real-time readouts of their brain activity and learn which mental strategies push it in desired directions. Lubianiker’s team split participants into three groups: one trained to upregulate the reward mesolimbic network (VTA and nucleus accumbens), another targeted non-reward brain regions as controls, and a third received no training at all. The twist? Neither participants nor experimenters knew who was in which group throughout the study, maintaining the double-blind design.
During each training session, participants chose mental strategies: recalling happy memories, imagining future rewards, visualizing sensory experiences. Then they received numerical feedback (1 to 10, with a smiley face that grew happier with better performance) showing how successfully they’d activated their target regions. Across three to four sessions, they refined their approaches, discovering which inner experiences most effectively drove their neural activity.
Immediately after the final session came the immune challenge: a hepatitis B vaccine. Blood samples at baseline, then 14 and 28 days post-vaccination, measured antibody responses.
The results were striking. Both neurofeedback groups showed increased VTA activity during training, which makes sense since neurofeedback itself involves reward-learning processes. But only VTA upregulation specifically correlated with antibody production (r = 0.31, P = 0.018). Nucleus accumbens activity? No correlation. Non-reward control regions? Nothing. The effect appeared three months later as well, though with fewer participants the correlation didn’t quite reach statistical significance.
The research team systematically ruled out alternative explanations. General neurofeedback success didn’t predict antibody levels. Neither did reward responsivity measured during feedback epochs, trait motivation, or personality measures. The link was specific to VTA regulation during the active upregulation periods.
The Psychology of Immunological Hope
What exactly were participants doing mentally when they successfully boosted VTA activity? The researchers developed a novel questionnaire to capture this. They had participants characterize each mental strategy across 45 features: sensory modalities, emotional content, cognitive aspects.
One feature stood out: positive expectation, defined as experiencing a positive expectation or excitement toward events or occurrences. At the start of training, positive expectations produced transient VTA activity that dissipated quickly. But by the final session before vaccination, participants who employed positive expectations showed sustained VTA upregulation throughout the 40-second regulation period.
This wasn’t merely about feeling happy or experiencing pleasure. Those features showed no such effect. It was specifically about expectation, about anticipation, about that forward-looking optimism we might colloquially call hope.
The pattern emerged uniquely in the VTA. While positive expectations triggered brief activity bursts in the nucleus accumbens and control regions, only VTA activity became coupled with sustained upregulation through training. The brain appeared to learn how to translate conscious positive expectations into prolonged reward signals.
Implications and Mechanisms
The work raises as many questions as it answers. How exactly does VTA activity reach out to influence peripheral immune cells? The classical routes (the hypothalamic-pituitary-adrenal axis, autonomic nervous system outflow) are obvious candidates. But recent work suggests the brain’s border tissues may matter too. Meningeal lymphatic vessels, which modulate both brain circuits and cognitive behavior, could serve as additional neuroimmune interfaces.
Jonathan Kipnis and colleagues at Washington University in St Louis, writing in an accompanying commentary, propose what they call a reward brain-meninges-immune axis. In this model, reward-related circuits engage meningeal functions that ultimately support peripheral immunity. Whether this pathway exists, and how it might operate, remains speculation for now.
The potential applications are tantalizing. Cancer patients receiving immunotherapy often suffer from depression, which disrupts reward circuitry. Could neurofeedback-based mesolimbic stimulation enhance therapeutic outcomes when combined with checkpoint inhibitors or CAR-T cell therapies? We’ll need rigorous clinical trials to evaluate this possibility.
There’s also the matter of scale. This was a mechanistic study with 85 participants, designed to demonstrate proof-of-concept rather than clinical efficacy. Post-vaccination antibody levels didn’t differ between groups, which makes sense given that both neurofeedback groups showed VTA activation. A larger trial specifically targeting VTA (rather than the broader mesolimbic network) might yield group-level differences.
The Placebo Reconsidered
Perhaps most profoundly, this work offers neural and immunological underpinnings for something physicians have long observed: positive expectations improve outcomes. Doctors have documented placebo effects extensively, yet the mechanisms remained murky. Now there’s imaging evidence that consciously generated expectations can engage specific reward circuitry, and that this neural activity correlates with measurable immune changes.
It’s not magic. It’s not wishful thinking defeating disease through sheer willpower. It’s a concrete biological pathway (albeit one researchers are only beginning to map) linking conscious mental states to peripheral physiology.
George Mashour’s earlier work on consciousness offers an apt parallel: consciousness is not something we see, it’s something through which we see. The same might apply to the reward system. It’s not just responding to external stimuli like mates, food, or safety. It’s actively constructing our body’s readiness to face challenges, infectious or otherwise.
The brain (that 1.4-kilogram mass of lipids and proteins generating our inner experience) constantly sends signals that shape immune cell behavior, hormone release, and inflammatory responses. We’ve thought of mind and body as separate for so long that demonstrating their integration still feels surprising. But the evidence keeps mounting.
Can hope heal? Not by itself, and not magically. But properly directed, it might shift our physiology in measurable ways. The volunteers lying in those fMRI scanners, mentally rehearsing positive expectations, weren’t just imagining better futures. They were, in a very real sense, building them.
Note: The human trial is registered as NCT03951870 at ClinicalTrials.gov
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.