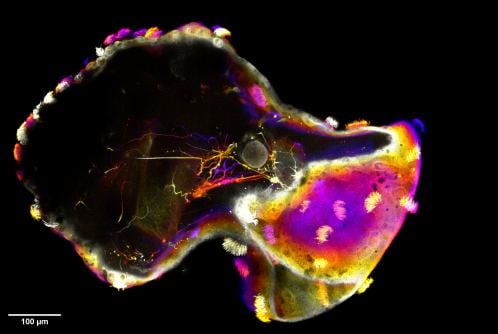
Slice away a small cap of cells from a frog embryo, drop them in a dish, and within hours something happens that still catches biologists off guard. The cells heal themselves into a tiny sphere, sprout hair-like cilia across their surface, and start swimming. No scaffold, no genetic tinkering, no instructions from a researcher. Just cells doing what cells apparently do when you take them out of the organism they were meant to build and leave them to sort things out on their own.
These living spheres, called xenobots (after the African clawed frog Xenopus laevis that donates the tissue), have been around since 2020. They can navigate watery environments, repair injuries, even gather loose cells and assemble copies of themselves. But a team at Tufts University and the Wyss Institute has now pushed the idea considerably further, implanting neural precursor cells into the developing spheres and watching what the neurons do when they wake up inside a body that evolution never designed. The resulting creatures, which the researchers call neurobots, are reported in Advanced Science.
Building one is a fiddly bit of microsurgery. You dissociate cells from roughly 50 frog embryos, keep them separated for about three hours (long enough that they commit to becoming neurons rather than skin), then reaggregate the cells into small clumps. While a freshly excised animal cap is still healing into its bowl shape, you tuck one or more of these neural clumps inside before the tissue seals shut. Within a couple of days the composite has healed, and by day three, cilia appear on the surface and the thing begins to move.
What the neurons get up to inside is, arguably, the most striking part. Staining with antibodies that bind to proteins found in mature nerve cells revealed that the implanted precursors had differentiated into proper neurons, extending axons and dendrites throughout the interior. Some of those projections reached all the way to the outer surface, making contact with the ciliated cells that drive locomotion. Protein markers associated with synapses turned up as well, scattered along the neural branches. Calcium imaging confirmed that the neurons were electrically active and, in some cases, firing in loosely coordinated patterns across distant regions of the construct.
The neurobots looked different from their nerveless siblings too. By day six they were significantly larger and more elongated (a sham control, implanted with cells that hadn’t been left long enough to adopt a neural fate, showed neither change). And they moved differently. While ordinary biobots tend to swim in simple circles or sit still for long stretches, neurobots were less likely to stop moving at all, and their trajectories were more complex, sometimes tracing spirograph-like repeating patterns across the dish. A spectral analysis of the movement data confirmed a statistically significant increase in what the researchers termed trajectory complexity.
“I’ve tried to understand neuronal behavior in existing animals like zebrafish, and how they give rise to behavior, but neurobots are about reverse engineering,” said Haleh Fotowat, senior scientist at the Wyss Institute. “Can we build a nervous system from the start? What happens if you put neurons in a completely novel context? What are the basic, innate rules for them to organize and form networks?”
To test whether neural signalling was genuinely behind the behavioural shift, the team dosed both biobots and neurobots with pentylenetetrazole, a drug known to block GABA receptors and induce seizures in animals. Most biobots responded by simplifying their movement patterns, probably because the drug was acting on GABA receptors found on mucus-secreting goblet cells lining the surface rather than on neurons (which biobots don’t have). Neurobots, on the other hand, responded in a significantly different way: the majority increased their movement complexity. Because neurobots are essentially biobots plus neurons, that divergence points to the nascent nervous system pushing back against the drug’s default inhibitory effect on the body. No two neurobots showed the same neural architecture under the microscope, which perhaps explains why a few did buck the trend, but the population-level difference was clear.
The transcriptomics data, though, is where things get properly weird. Bulk RNA sequencing showed that neurobots had massively upregulated gene expression compared to both biobots and shams, with nearly 6,800 genes significantly more active. The expected categories were there: synapse formation, neurotransmitter receptors (glutamate, GABA, dopamine, serotonin, acetylcholine, glycine), voltage-gated ion channels, axon growth. But one cluster stood out. Genes involved in visual perception, phototransduction, and photoreceptor development had switched on. Cone opsins, rhodopsin, melanopsin, retinal G-protein coupled receptors, and genes found in specific retinal cell types including ganglion cells and horizontal cells were all significantly upregulated. In a creature with no eyes, no evolutionary history of seeing, and roughly nine days to live.
“We don’t know, but my hypothesis is that these neurobots are up-regulating parts of the genome that could be useful for novel functions down the line,” said Michael Levin, director of the Allen Discovery Center at Tufts. “If they lived longer, would they then also develop photoreceptors? It’s a fascinating question that we are actively studying.”
A phylostratigraphic analysis added another layer: more than 54 percent of the genes upregulated in neurobots fell into the two most ancient categories, shared across all living organisms or across eukaryotes broadly. The construct’s transcriptome, in other words, was shifting toward a very old genetic programme, as though assembling a nervous system from scratch meant reaching back to the evolutionary basement for parts. Gene expression variability between individual neurobots was also significantly higher than between biobots, which the researchers suspect reflects both the manual variability of construction and the neurons themselves driving exploratory changes in gene expression across the construct.
Where all of this leads is still genuinely open. The team wants to know whether the visual-perception genes translate into actual proteins, whether light can modulate motor output, whether optogenetic tools could establish causal links between neural firing and ciliary beating. If neurobots could one day sense and respond to light, that would be a wholly emergent behaviour, something these cells were never selected to do in this configuration. For now, the nervous systems growing inside these tiny frog-cell spheres are already rewriting rules about what neurons will build when you give them a body and get out of the way.
DOI / Source: https://doi.org/10.1002/advs.202508967
Frequently Asked Questions
What exactly is a neurobot, and how is it different from a xenobot? A neurobot is a small living construct made from frog embryo cells, like a xenobot, but with neural precursor cells implanted during its formation. Those precursors mature into functional neurons that wire themselves throughout the body. The key difference is that neurobots develop a primitive self-organized nervous system, which changes their shape, size, and movement patterns compared to nerveless xenobots.
Can neurobots actually see? Not yet, and possibly not ever in their current form. What researchers found is that genes associated with visual perception, including opsins and retinal cell-type markers, are actively switched on in neurobots despite the absence of anything resembling an eye. Whether those genes produce functional proteins or influence behavior remains an open question that the team is now investigating.
Why did the neurons start building themselves into a nervous system without any instructions? Neural precursor cells carry an innate capacity for self-organization that doesn’t require external patterning signals or genetic manipulation. When placed inside the healing frog-cell sphere, they differentiated into mature neurons, extended axons and dendrites, and formed putative synaptic connections on their own. The research suggests that neurons have built-in rules for network formation that operate even in a body plan evolution never tested.
Could this research lead to medical applications? The long-term goal includes insights for regenerative medicine and synthetic biology. Understanding how neurons self-organize in novel contexts could inform strategies for restoring damaged nervous tissue or building innervated biological constructs. However, the work is still at a fundamental-science stage, and clinical applications remain distant.
How long do neurobots survive? About 9 to 10 days, the same lifespan as ordinary xenobots. They aren’t fed externally but sustain themselves on yolk platelets stored in the original embryonic cells. One of the open questions is whether extending their lifespan could allow the activated visual-perception genes to produce more complex sensory structures.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.