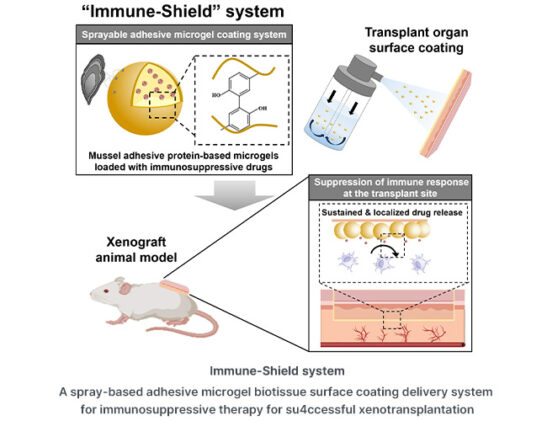
The bottle looks ordinary enough. Press the nozzle, and a fine mist settles over a glistening organ surface, disappearing almost instantly into the wet tissue. What it leaves behind, invisible to the naked eye, is perhaps the most clinically useful thing: a coat of microscopic drug-loaded beads that grip the tissue like a mussel grips wet rock, and release their payload slowly, over weeks, directly where the immune system is most likely to attack.
The idea that organ transplantation still runs on the same basic pharmacological logic it did in the 1970s surprises most people. Patients receive cyclosporin A or similar calcineurin inhibitors by mouth or intravenous drip, the drugs flood the bloodstream, and the immune system is blunted everywhere at once. It works, roughly. But kidneys suffer, infection risk climbs, and the cumulative toll of decades on systemic immunosuppression falls entirely on the person the drugs were meant to protect.
A team at Pohang University of Science and Technology (POSTECH) and Ewha Womans University, led by chemical engineer Hyung Joon Cha, has been trying to make that logic obsolete. Their system, which they call Immune-Shield, uses protein microgels inspired by the adhesive chemistry of mussels to deliver cyclosporin A exclusively to the graft site. Wet surface adhesion, sustained drug release, and a protein architecture that the immune system barely notices. The results, published in the Journal of Controlled Release, suggest the approach could significantly change what immunosuppression looks like after a transplant.
The mussel connection is not decorative. Marine mussels attach to rocks, pier pylons, and ship hulls through a combination of positively charged lysine residues and tyrosine-based chemistry that forms stable bonds under water, conditions that defeat most synthetic adhesives. Cha’s group has spent years engineering bacterial versions of the relevant mussel foot proteins, fp-1 and fp-5, and the resulting protein has roughly 20 mol% lysine and a similar proportion of tyrosine, giving it much of the same wet-surface stickiness. Crosslink that protein into spherical microgels about nine micrometres across using a flash of visible light, load them with cyclosporin A, suspend them in an ethanol-saline mix that atomises cleanly under manual pump pressure, and you have a sprayable formulation that coats even the irregular contours of a transplanted organ in a few passes.
What happens next is the part that matters. In ex vivo testing on porcine skin, more than half the microgels were still detectable after six days of daily rinsing in physiological buffer; in live mice, the same fraction persisted to day twelve before the protein matrix degraded entirely. Non-adhesive control microgels, made from bovine serum albumin rather than the mussel protein, were gone within a day. The drug itself was released over roughly seventeen days, with nearly 90 percent of the cyclosporin A distributed by that point, following a diffusion and degradation profile that the team fitted to the Korsmeyer-Peppas kinetic model. That kind of sustained release is precisely what a graft needs in the critical early weeks, when rejection pressure is highest and systemic drug dosing carries the most risk.
The practical test used a xenograft model in which skin tissue from Sprague-Dawley rats was transplanted onto immunocompetent BALB/c mice. It is, admittedly, a long distance from the clinical context of human organ transplantation. But the immunological challenge is real: the mouse immune system recognises the rat tissue as foreign, mounts a T cell-mediated response, and in untreated animals the plasma levels of interleukin-2 and interferon-gamma rise to roughly twice normal within a week. Cha’s team sprayed the graft with either free cyclosporin A solution or Immune-Shield at the same drug dose before suturing it in place, then measured immune markers on days seven and twenty-one.
At seven days, both treatments had suppressed the IL-2 response. By day twenty-one, the free-drug group had essentially lost that advantage: its cytokine levels were comparable to untreated xenograft animals, the drug having dispersed before any continued effect could be sustained. The Immune-Shield group, by contrast, showed less than half the IL-2 concentration of untreated controls at the same timepoint. CD8-positive cytotoxic T cell infiltration at the graft site was also markedly lower, and haematoxylin and eosin staining of the tissue confirmed reduced inflammation. Same drug, same initial dose; the delivery mechanism accounted for the difference.
“We propose a strategy to solve the long-standing challenge of immunosuppressants by using mussel adhesive protein, an original biomaterial developed in Korea,” Cha said. The framing of that statement contains an ambition: that locally targeted immunosuppression, rather than systemic suppression, is the right direction for the field. There is decent evidence to support the intuition. Localised drug delivery systems avoid the off-target toxicity that accumulates with years of calcineurin inhibitor use, and they can maintain higher drug concentrations at the site where they are actually needed without those concentrations propagating through the bloodstream to kidneys, liver, and bone marrow.
Whether the approach scales to the full complexity of solid organ transplantation is a different question, and Cha’s group is careful about it. The skin xenograft model demonstrates principle, not clinical readiness. Pharmacokinetic studies and systemic toxicology assessments remain to be done. Internal organs with irregular geometry present coating challenges that flat skin tissue does not, though the team notes that the spray system’s conformability is an advantage here. “Since the spray method allows for easy application to complex organ surfaces, it is expected to serve as a key technology to increase the success rate in the field of xenograft transplantation in the future,” Cha said.
The protein itself has a quality that turns out to be useful in an immunosuppressive context. The bioengineered mussel adhesive protein is intrinsically disordered, meaning it lacks stable secondary structure, and intrinsically disordered proteins tend to elicit weak immune responses, possibly because the immune system has a harder time recognising and mounting an attack on something that doesn’t hold a fixed shape. Loading that low-immunogenicity scaffold with a drug meant to suppress the immune system has a certain elegance to it: the carrier avoids triggering the very response the drug is intended to control.
Xenotransplantation, the transplantation of animal organs into humans, remains one of the more logistically fraught problems in medicine. The organ shortage is real and growing; demand far outpaces supply from human donors. The immunological hurdles involved in crossing species lines are correspondingly larger than those in allotransplantation. A localised sustained-release coating that could buy time for the immune system to accommodate a foreign graft, rather than hammering it into submission systemically, might eventually be one component of what makes the whole enterprise more viable.
Study link: https://www.sciencedirect.com/science/article/pii/S016836592501082X
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.