Researchers have discovered a remarkable peptide derived from natural human proteins that fights viruses while simultaneously promoting tissue regeneration and wound healing.
The breakthrough substance, called Ac-Tβ1-17, demonstrated over 85% inhibition of a key COVID-19 virus enzyme while accelerating cell growth, blood vessel formation, and healing processes in laboratory studies—potentially revolutionizing how we approach both infectious diseases and regenerative medicine.
The peptide emerges from the breakdown of thymosin β4, a protein naturally present throughout the human body. Unlike traditional therapeutics that target single problems, this metabolite tackles multiple health challenges simultaneously, offering hope for more versatile and effective treatments.
From Protein Breakdown to Medical Breakthrough
The discovery originated from research into peptide metabolites—small fragments produced when larger proteins break down naturally in the body. These molecular remnants, often overlooked in drug development, may harbor therapeutic potential that surpasses their parent proteins.
“This study demonstrates that protein metabolites can be used not only as new drugs but also as biomaterials for tissue regeneration, confirming their potential for expansion into various biomedical applications,” explained Dr. Hyung-Seop Han from the Korea Institute of Science and Technology’s Biomaterials Research Center.
The research team, led by Dr. Han alongside colleagues Dr. Dae-Geun Song and Dr. Oh-Seung Kwon, identified Ac-Tβ1-17 through systematic screening of thymosin β4 metabolites. Their analysis revealed 13 different metabolic fragments, but Ac-Tβ1-17 stood out for its exceptional biological activity.
Dual-Action Against Disease and Damage
Laboratory testing revealed Ac-Tβ1-17’s impressive versatility across multiple therapeutic areas:
- Antiviral activity: 85.77% inhibition of SARS-CoV-2’s main protease (Mpro) versus only 7.09% for the parent protein
- Wound healing: Significantly enhanced closure rates in cell culture studies
- Blood vessel formation: Promoted angiogenesis crucial for tissue repair
- Cellular protection: Scavenged harmful reactive oxygen species
- Growth promotion: Increased cell proliferation and migration
The peptide’s antiviral mechanism involves binding to COVID-19’s main protease, a crucial enzyme for viral replication. Detailed molecular analysis showed the peptide’s C-terminal region forms specific bonds with the enzyme’s active site, effectively blocking viral function.
Engineering Therapeutic Scaffolds
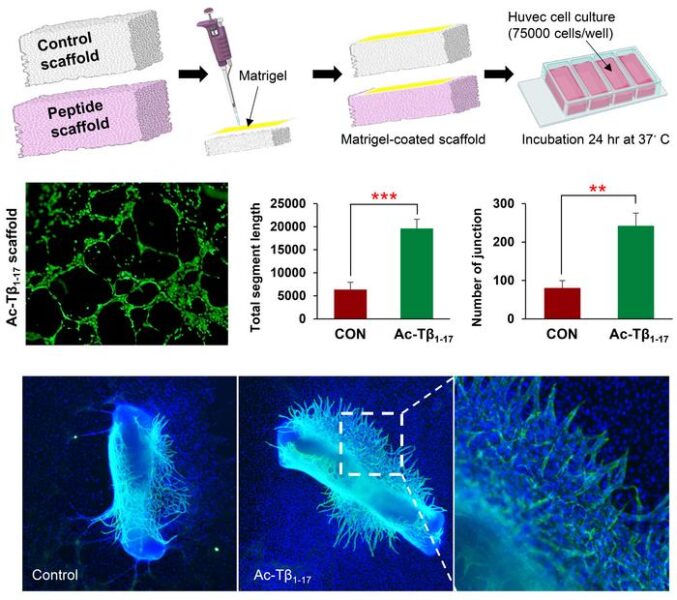
Beyond its direct therapeutic effects, the researchers successfully incorporated Ac-Tβ1-17 into biomaterial scaffolds—structural frameworks that support tissue growth and repair. These peptide-enhanced scaffolds maintained the molecule’s biological activity while providing sustained release over several days.
When human vascular cells were cultured on these scaffolds, researchers observed enhanced cell adhesion, proliferation, and blood vessel formation compared to control materials. The scaffolds’ porous structure allowed gradual peptide release while supporting cellular growth throughout the matrix.
Importantly, the peptide retained its activity even after scaffold preparation at high temperatures (80-90°C), an advantage over protein-based growth factors that typically degrade under such conditions.
Molecular Mechanisms Revealed
Comprehensive analysis of Ac-Tβ1-17’s effects on human umbilical vein endothelial cells revealed the peptide’s sophisticated molecular mechanisms. Treatment significantly upregulated genes crucial for cell survival and growth, including AKT, ERK, PI3K, MEK, and Bcl-2.
Protein expression studies identified 18 significantly upregulated and four downregulated proteins following treatment. Many of these proteins participate in critical cellular pathways including the EGFR-VEGFR cascade, fundamental to blood vessel development and tissue repair.
The peptide also influenced the balance between cell death and survival signals, reducing the BAX/Bcl-2 ratio to favor cellular survival—essential for effective tissue regeneration.
From Laboratory to Clinical Promise
To validate their findings beyond cell culture, researchers tested Ac-Tβ1-17 in fetal mouse metatarsal bone explants, a well-established model for studying blood vessel formation. Five-day treatment significantly increased both bone size and vascular network development, with enhanced vessel density, branching, and connectivity.
Safety testing revealed no concerning toxicity in various cell types or red blood cells, even at concentrations up to 1000 μg/mL. This favorable safety profile reflects the peptide’s natural origin from human proteins.
The research addresses growing interest in peptide-based therapeutics, particularly following recent successes like Wegovy for weight management. However, most current peptide drugs target single conditions, making Ac-Tβ1-17’s multifunctional capabilities particularly noteworthy.
A New Paradigm for Drug Discovery
The work represents a paradigm shift in therapeutic development, suggesting researchers should examine protein metabolites more closely rather than focusing solely on intact proteins. Natural breakdown products may offer enhanced or novel biological activities compared to their precursors.
“We will continue research using natural bioactive materials to pursue practical applications in antivirals, functional biomaterials, and beyond,” noted Dr. Song from the research team.
This approach could prove especially valuable given the safety advantages of naturally-occurring metabolites. Since these fragments normally appear in healthy human bodies, they may face fewer regulatory hurdles and exhibit better biocompatibility than synthetic alternatives.
Future Applications and Challenges
The researchers envision diverse applications for Ac-Tβ1-17, from standalone antiviral treatments to sophisticated tissue engineering scaffolds. The peptide’s dual functionality could prove particularly valuable in treating complex conditions where both infection control and tissue repair are needed.
However, several challenges remain before clinical application. Researchers must determine optimal dosing regimens, delivery methods, and potential side effects in whole organisms. The peptide’s stability in biological systems and manufacturing scalability also require investigation.
Dr. Kwon emphasized the collaborative nature of the breakthrough: “The metabolite of thymosin β4 has been identified as a drug candidate through collaborative research, and we expect it to be widely applicable in this field moving forward.”
As the team continues developing practical applications, their work opens new avenues for exploring how nature’s own molecular recycling processes might yield tomorrow’s most effective medicines. In an era where single-target drugs often fall short of complex medical needs, multifunctional molecules like Ac-Tβ1-17 may represent the future of therapeutic innovation.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.