What if your body could manufacture its own weight-loss medication, eliminating the need for weekly injections that cost hundreds of dollars and often lead to rebound weight gain when stopped?
Japanese researchers have engineered a single genome editing treatment that transforms liver cells into biological drug factories, producing sustained levels of appetite-suppressing hormones for months.
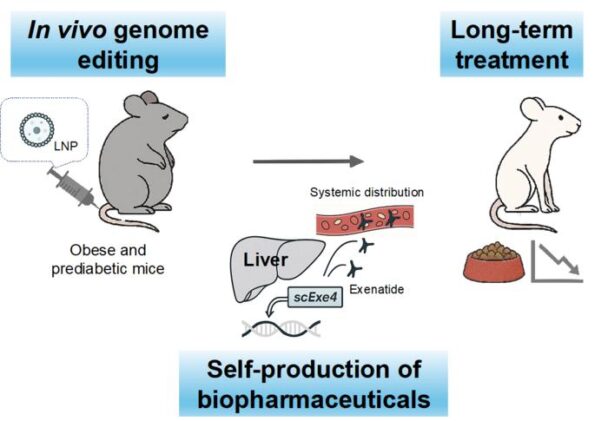
The approach, published in Communications Medicine, represents a paradigm shift from traditional genome editing, which corrects genetic mutations, to creating entirely new biological functions. Instead of fixing broken genes, the technique adds new ones—turning the liver into a pharmaceutical manufacturing plant.
Engineering Biological Drug Production
The University of Osaka team targeted Exenatide, a GLP-1 receptor agonist that mimics hormones naturally produced after eating. These medications signal fullness to the brain and slow digestion, leading to reduced appetite and weight loss. However, like most protein-based drugs, Exenatide breaks down quickly in the body.
“An alternative to genome editing for many complex and non-genetic diseases is biologic medications, which are essentially injectable proteins,” explains senior author Keiichiro Suzuki. “These medications do not stay in the body long, meaning they typically have to be injected weekly, or even daily, to maintain consistent therapeutic levels of the drug.”
The researchers solved this problem by engineering a modified version of Exenatide—called scExenatide—designed for continuous secretion. They fused the drug with cellular “shipping tags” that direct proteins out of cells and added enzymes that process the medication into its active form once secreted.
Precision Targeting and Sustained Results
Using a technique called HITI (Homology-independent target integration), the team inserted the scExenatide gene directly into the albumin locus—a highly active gene region in liver cells. This strategic placement ensures robust, liver-specific production of the weight-loss compound.
The results proved remarkable. In obese, pre-diabetic mice, a single treatment maintained detectable Exenatide levels in the bloodstream for 28 weeks—the entire duration of the study. This sustained drug production translated into significant metabolic benefits:
- 29% reduction in food intake compared to untreated obese mice
- 34% decrease in body weight, matching levels seen in lean control mice
- Improved glucose tolerance and insulin sensitivity
- Normalized blood sugar markers including HbA1c levels
Advantages Over Current Treatments
The genome editing approach demonstrated clear advantages over conventional drug delivery. While mice receiving continuous Exenatide via implanted pumps showed benefits only during active treatment, the genome-edited animals maintained their improvements throughout the study period.
When researchers removed the drug pumps at 8 weeks to simulate treatment interruption, the control mice quickly regained weight and lost metabolic benefits. The genome-edited mice, however, continued producing their own medication and sustained their weight loss.
Digital PCR analysis revealed that only about 1% of liver cells successfully integrated the new gene—yet this modest efficiency proved sufficient for therapeutic effect. The high expression level of the albumin gene compensated for the relatively low number of modified cells.
Safety and Broader Applications
Safety analyses showed no significant liver toxicity or off-target genetic effects. The treatment didn’t interfere with natural hormone production—endogenous GLP-1 levels remained normal even with continuous Exenatide secretion.
The researchers used lipid nanoparticles similar to those in approved therapies to deliver the genome editing components specifically to liver cells. This targeted approach minimized systemic exposure and potential side effects.
“We hope that our design of a one-time genetic treatment can be applied to many conditions that do not have exact genetic causes,” Suzuki notes. The technique could potentially work for other protein-based medications used to treat inflammatory diseases, autoimmune conditions, and metabolic disorders.
Future Implications
The approach addresses a fundamental limitation of current biologics—their short half-lives requiring frequent dosing. By converting the body into a drug manufacturing facility, the technique could improve treatment adherence, reduce costs, and prevent the rebound effects common when medications are discontinued.
However, questions remain about long-term safety, optimal dosing, and patient selection. The researchers acknowledge that future studies must evaluate the approach in diverse obesity models and assess effects on gastric emptying and other physiological functions.
This work opens new possibilities for treating complex diseases without clear genetic causes—conditions that affect millions but have remained largely beyond the reach of traditional gene therapy. If successfully translated to humans, it could transform how we approach chronic disease management from repeated treatments to one-time interventions.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.