Pancreatic tumors thrive on a self-reinforcing loop. Three cancer-driving genes lock together in a circuit that keeps feeding itself, pushing cells to divide faster and resisting most treatments. Cold Spring Harbor Laboratory researchers have now found a way to break that loop by targeting not the genes themselves, but how one of them gets processed into working protein.
The work, published in Molecular Cell, focuses on pancreatic ductal adenocarcinoma, the most common and deadliest form of the disease. Rather than attacking the notorious KRAS mutation directly, a strategy that has repeatedly failed, the team went after the RNA splicing machinery that tumors depend on to assemble their growth engines.
The Circuit That Won’t Stop
The loop involves three major oncogenes. SRSF1, an RNA splicing protein, alters how Aurora kinase A (AURKA) gets built, boosting its production. AURKA then stabilizes MYC, a powerful gene that drives cell division. MYC pushes SRSF1 levels back up, restarting the cycle.
Alexander Kral, a former graduate student in Adrian Krainer’s lab, spotted the pattern while revisiting earlier data showing that SRSF1 could jumpstart pancreatic tumors. “Our theory was that some of the changes caused by increased levels of SRSF1 were playing a role in the accelerated tumor growth we were seeing,” Kral explains. “We homed in on a molecule we thought could be an important driver of this called Aurora kinase A.”
Parts of this circuit were known before, but the full picture remained unclear until the team discovered that alternative splicing of AURKA was the linchpin. That realization opened a path to disruption.
The researchers designed an antisense oligonucleotide, a short synthetic RNA strand, to interfere with AURKA splicing. The Krainer lab has deep experience here. They previously developed Spinraza, the first FDA-approved treatment for spinal muscular atrophy, using the same approach.
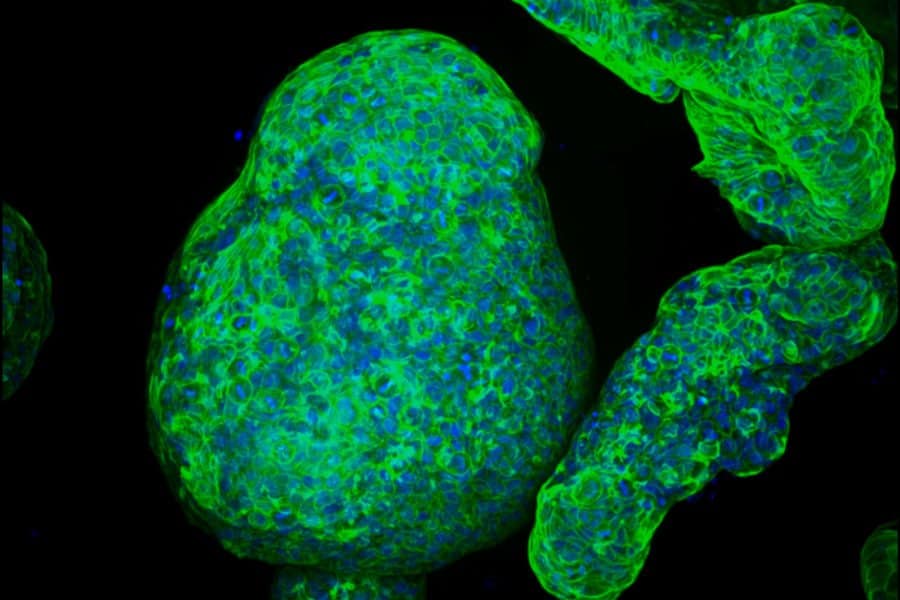
When tested in human pancreatic tumor organoids, three-dimensional lab-grown models of patient tumors, the ASO caused the entire circuit to fail. Cancer cell viability dropped. Apoptosis, programmed cell death, kicked in.
“It’s like killing three birds with one stone,” Krainer explains. “SRSF1, AURKA, and MYC are all oncogenes contributing to PDAC progression. Just by targeting AURKA splicing with our ASO, we see the loss of these other two molecules as well.”
An Evolutionary Quirk Turned Cancer Driver
The team also uncovered something unexpected about the genetic segment their ASO targets. It comes from Alu elements, ancient primate-specific sequences embedded in human DNA. In healthy tissue, cells ignore these Alu-derived segments during RNA processing. In pancreatic cancer, SRSF1 forces the cell to include them, and that inclusion drives aggressive tumor growth.
Clinical use remains years away. The ASO needs refinement and extensive testing before it could reach patients. But the work reframes how to think about pancreatic cancer treatment. Instead of chasing single mutations, the strategy focuses on dismantling the internal logic tumors use to sustain themselves.
For Krainer, the trajectory feels familiar. Spinraza also began as basic science about RNA splicing before changing thousands of lives. If this approach follows a similar path, it could crack open a cancer that has long seemed impenetrable.
Molecular Cell: 10.1016/j.molcel.2025.12.004
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.