Three sulfur atoms in a row. That is, more or less, the entire secret. The trisulfide bond, a structural feature found in everything from garlic to anti-cancer drugs to vulcanized rubber, turns out to behave in a way that chemists have somehow missed for more than a century of study: dissolve it in the right solvent and it spontaneously swaps its atomic partners, trading and reconnecting at room temperature, without heat, without light, without any catalyst at all, reaching equilibrium within seconds in some cases.
The discovery, published this week in Nature Chemistry, was not exactly the result of a targeted hunt. It emerged from something more like a controlled surprise, when researchers at Flinders University in Adelaide noticed that sulfur polymers they were studying were rearranging themselves in ways the theory of the day couldn’t account for.
Sulfur-sulfur bonds are ubiquitous in chemistry. They hold protein structures together, they appear in drug molecules, they crosslink the long polymer chains in rubber. Chemists have known for decades that these bonds can be made to break and reform, which is useful: self-healing materials, drug delivery systems, and combinatorial drug libraries all exploit this property. The problem has always been that provoking the exchange requires something. Heat, typically, or ultraviolet light, or a nucleophilic catalyst: some external shove to get the reaction started. And because that shove is rarely precise, the chemistry tends to produce a messy mixture of different bond lengths: disulfides (two sulfurs), trisulfides (three), tetrasulfides (four), and more.
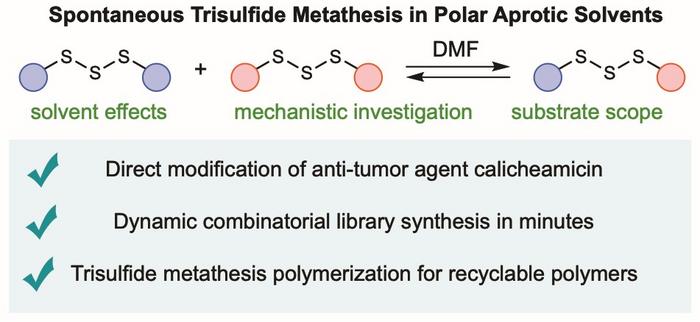
The new reaction (which the team calls trisulfide metathesis) does something quite different. It is exquisitely selective.
When organic trisulfides are placed in a polar aprotic solvent, a category that includes common laboratory solvents like dimethylformamide (DMF) and dimethylsulfoxide (DMSO), the S-S-S bonds begin spontaneously exchanging their organic groups with each other. Within minutes at room temperature, or seconds if the solvent concentration is high enough, an equilibrium is established. And crucially, only trisulfides are produced; no disulfides, no tetrasulfides, none of the contaminating byproducts that normally accompany sulfur chemistry. The selectivity is, by the standards of sulfur exchange reactions, rather remarkable.
“It is rare to discover an entirely new reaction, and even more rare for it to be useful in so many fields and applications,” said Professor Justin Chalker, who led the research at Flinders University and has spent more than a decade working on environmentally friendlier sulfur polymers.
What makes the selectivity possible is, the team believes, a matter of mechanism. Standard sulfur exchange reactions proceed through either radical intermediates or ionic ones, both of which generate that cocktail of different polysulfides. The trisulfide metathesis, by contrast, seems to involve a transient species called a thiosulfoxide: a non-linear rearrangement of the trisulfide containing a double bond between two sulfur atoms. This reactive intermediate, predicted by the team’s computational work to sit roughly 67 kilojoules per mole higher in energy than the original trisulfide, is polar enough to be stabilised in polar solvents, explaining why the reaction simply does not occur in hydrocarbons or alcohols. The selectivity falls out naturally: the thiosulfoxide can only make and break trisulfide bonds, not disulfide or tetrasulfide ones.
That clean selectivity immediately suggested something to the researchers. A natural product called calicheamicin, one of the most potent anti-tumour compounds ever discovered, contains a trisulfide in the middle of a molecule bristling with other reactive groups: an enediyne warhead, an unsaturated ketone, a thioester. Modifying calicheamicin selectively has historically been something of a nightmare; existing methods tend to disturb the rest of the molecule, setting off a cascade of unwanted reactions including, in the worst cases, triggering the enediyne to cyclise and destroy the drug. The team treated calicheamicin with excess dibenzyl trisulfide in DMF and watched, by NMR spectroscopy, as it underwent clean trisulfide metathesis in around ten minutes, with none of the other reactive groups disturbed. It is the kind of selectivity medicinal chemists tend to plump for rather wistfully in meetings.
The polymer chemistry is perhaps equally striking. Using the same exchange reaction but in reverse, the researchers polymerised a bifunctional trisulfide monomer into a long-chain material with properties similar to high-density polyethylene. The polymer can be injection-moulded into solid objects. What distinguishes it from conventional polyethylene is that adding a small amount of dimethyl trisulfide in DMF causes the whole structure to depolymerise back to starting monomer in under five minutes, with recovery yields of around 91 percent. Closed-loop chemical recycling. No degradation of the feedstock.
The recycling idea is not entirely new; there has been a growing effort over the past few years to design polymers that can be unmade on demand. What the trisulfide metathesis adds is simplicity. No specialised catalyst, no high-temperature reactor, no exotic reagents. Just a solvent and a small trigger molecule.
Where this goes next is genuinely hard to predict. Dr Tom Hasell, a Royal Society University Research Fellow at the University of Liverpool and a collaborator on the study, noted that the demonstrated applications represent “only the tip of the iceberg.” New ARC Discovery Grant funding in Australia will expand the chemistry to recyclable rubbers, foams, and fibres. The drug-modification angle seems likely to attract interest from antibody-drug conjugate developers, who routinely struggle with exactly the kind of selective trisulfide manipulation this chemistry enables. And the dynamic combinatorial library work, in which a mixture of eight trisulfides spontaneously generated all 29 possible permutations within five minutes of adding DMF, opens up a route to rapid drug discovery that sidesteps some of the fussier pH-sensitivity problems that plague existing disulfide exchange systems.
The reaction has been sitting there, in the solvent, for as long as people have been doing sulfur chemistry. Nobody had quite asked the right question of the right compound in the right conditions to notice it.
DOI / Source: https://doi.org/10.1038/s41557-026-02091-z
Frequently Asked Questions
Anti-cancer drugs like calicheamicin contain trisulfide bonds that researchers need to modify precisely in order to attach them to antibodies for targeted delivery. Until now, manipulating that bond without disturbing the rest of the molecule was extremely difficult, because available chemistry tended to trigger unwanted side reactions. The trisulfide metathesis reaction is selective enough to modify only the trisulfide, leaving other reactive groups intact, which could meaningfully expand what’s possible in antibody-drug conjugate design.
Normally, making sulfur-sulfur bonds exchange requires some kind of external push: heat, light, or a chemical catalyst. And the result is typically a messy mixture of bonds of different lengths. The new reaction happens spontaneously at room temperature in certain common solvents, and it produces only trisulfide products, no disulfides or tetrasulfides. That combination of spontaneity and selectivity is what makes it unusual enough to count as a genuinely new class of reaction.
Not on its own, but it demonstrates something genuinely useful: a polymer material with physical properties close to polyethylene that can be chemically depolymerised back to its original building blocks in under five minutes, with very high recovery. The chemistry is simple enough that it could in principle be scaled. Whether this becomes a practical route to recyclable commodity plastics depends on cost, durability, and whether the approach can be extended to the full range of polymer types people actually use.
A thiosulfoxide is an unstable rearrangement of a trisulfide in which two of the sulfur atoms form a double bond, creating a more reactive and more polar intermediate. The Flinders team’s computational modelling suggests this intermediate forms transiently in polar solvents and is responsible for the exchange reaction, which is why the chemistry works in dimethylformamide but not in hydrocarbons. Understanding the mechanism matters because it explains the selectivity and suggests which other substrates might work, or not.
The solvents involved, principally DMF and DMSO, are standard laboratory chemicals with well-understood handling requirements; neither is exotic or particularly hazardous by industrial standards. The reaction occurs at room temperature without pressure or high heat, which simplifies the safety picture considerably. Whether large-scale industrial use is feasible will depend on questions about solvent recovery, reaction volume, and how the chemistry scales, none of which have been fully addressed yet.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.
Key Takeaways
- The new trisulfide metathesis reaction enables sulfur atoms to exchange partners spontaneously in polar aprotic solvents at room temperature.
- This reaction avoids unwanted byproducts, producing only trisulfides and maintaining selectivity, unlike traditional sulfur chemistry.
- Potential applications include precise modifications of anti-cancer drugs like calicheamicin and creating recyclable polymer materials.
- The method simplifies chemical recycling without specialized catalysts or high temperatures, enabling high recovery yields.
- Future research may expand its applications, targeting sustainable practices in drug development and materials science.