Think about what happens inside a solar cell the moment light hits it. Photons jostle electrons loose from their atoms, and those electrons have to travel, quickly, through a crystalline lattice before they recombine and the energy is wasted as heat.
In a tandem cell built from two stacked layers of perovskite, a man-made mineral compound that can be tuned to absorb different wavelengths, this journey is complicated by a fundamental mismatch: the two layers crystallize at different speeds and in different ways, producing misaligned microstructures riddled with defects, and those defects act like traps, swallowing electrons before they reach the electrode. The physics is unforgiving. No amount of clever engineering at the device level can fully compensate for what goes wrong at the crystal level, where individual atoms bond, cluster, and arrange themselves in the fraction of a second after the wet ink of precursor solution hits the substrate.
That atomic-scale problem is the central obstacle between perovskite solar technology and the kind of efficiency numbers that would let it challenge silicon in commercial photovoltaic markets, and a team at the Ningbo Institute of Materials Technology and Engineering (NIMTE) of the Chinese Academy of Sciences thinks they have found a way around it.
A perovskite tandem cell stacks two light-absorbing layers on top of each other, each tuned to capture a different part of the solar spectrum. Because sunlight spans a wide range of wavelengths, a single layer can only efficiently convert a portion of it; stacking two layers (or more) allows more of that energy to be captured before it is lost as heat. Perovskite is the general name for a class of synthetic mineral compounds that can be chemically tuned to absorb different wavelengths, which makes them well-suited to this kind of stacked architecture.
When the liquid precursor solution dries and solidifies into a thin film, the way atoms arrange themselves determines how easily electrons can travel through the material. Imperfect crystals, with misaligned grains or undercoordinated atoms at their boundaries, act as traps for electrons; those trapped electrons can’t reach the electrode and the energy they carry is lost. Controlling crystallization, so that films grow more uniformly with fewer of these trapping sites, is one of the central engineering challenges in perovskite solar research.
Most previous strategies tried to correct crystallization defects after the film had already formed, through surface treatments or additional processing steps. The NIMTE team intervened much earlier, modifying the precursor solution itself before deposition, so that both perovskite layers grow more uniformly from the outset. The use of hard-soft acid-base theory to select molecules that target specific metal ions in each layer is a more deliberate and systematic approach than many earlier trial-and-error methods.
Several companies are working toward commercial perovskite modules, and some limited products have reached the market, but widespread commercial deployment is probably still several years away. The main remaining challenges are long-term stability (cells need to last 25 to 30 years in outdoor conditions), lead content restrictions in some regions, and the difficulty of maintaining high efficiency when manufacturing at large scale rather than in small laboratory batches. The efficiency numbers being achieved in research labs are already well above commercial silicon, so the barriers are largely engineering and regulatory rather than fundamental scientific ones.
All-perovskite tandems are theoretically cheaper to manufacture than perovskite-silicon hybrids because both layers can be deposited from solution rather than requiring the high-temperature processes used to make silicon. If the efficiency and stability challenges are solved, a fully solution-processable tandem cell with efficiencies above 30% could significantly reduce the cost of generating solar electricity, particularly in applications where panel weight or flexibility matters, such as building-integrated photovoltaics or portable power.
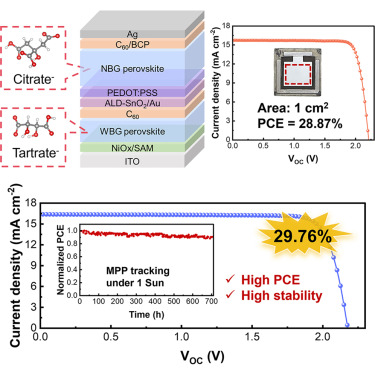
The approach, published in Joule on March 27, is built around a branch of chemistry that most solar researchers don’t typically reach for: colloidal science, the study of how tiny clusters of material suspended in a liquid behave before they solidify. Rather than trying to fix crystallization defects after the fact, with surface treatments or post-deposition anneals, the NIMTE group led by Professors GE Ziyi and LIU Chang chose to intervene much earlier in the process, reshaping the precursor solution itself so that both perovskite layers grow more uniformly from the start. The key turned out to be a pair of small, charged molecules, tartrate and citrate, chosen through a framework called hard-soft acid-base theory.
Hard-soft acid-base theory is, in a way, chemistry’s matchmaking system. It sorts metal ions and molecular ligands by how tightly they hold onto their electrons, and it predicts which pairs will bond strongly. The NIMTE team used it to pick two carboxylate anions with different electronic characters, each tuned to interact with a different perovskite layer.
In the top layer (the wide-bandgap subcell, which absorbs shorter-wavelength light), the problem is phase segregation: halide ions drift under illumination, creating regions with different chemical compositions, and efficiency collapses. Tartrate, a borderline-base anion, preferentially coordinates with lead ions in the precursor solution, stabilizing the clusters that form before the film solidifies and suppressing that drift. In the bottom layer (the narrow-bandgap subcell, built around a tin-lead mixture and tuned to harvest longer wavelengths), the problem is different: tin is badly behaved, oxidizing from Sn2+ to Sn4+ easily, and that oxidation creates a dense population of electronic defects that kill charge transport. Citrate, a harder-base anion, coordinates more strongly with Sn-I bonding networks in the precursor colloids, reducing the number of undercoordinated tin sites before the film even forms. A third component, choline cations, works at the crystal-colloid interface to mop up remaining undercoordinated metal ions in both layers.
The researchers note that this work “provides a universal approach to harmonize multijunction crystallization, paving the way for the commercialization of high-efficiency all-perovskite tandem solar technologies.” The language is careful and it should be: scaling any laboratory result to commercially viable manufacturing is genuinely hard, and perovskite in particular has a long track record of promising bench-top numbers that prove difficult to reproduce at larger sizes.
Which is why the team’s large-area result is probably the more significant figure in the paper. The headline number is 29.76% power conversion efficiency for a small tandem device (certified independently at 29.22%), but the 1 cm2 version, which is closer to what you’d actually need for a manufacturable module, came in at 28.87%. The gap between small-cell and large-cell performance is usually where perovskite optimism goes to die; a difference of less than one percentage point is unusually tight, and it suggests the colloidal chemistry approach may be doing something genuinely useful for uniformity at scale. Stability testing added to the picture: the device retained more than 90% of its initial efficiency after 700 hours of continuous operation under maximum power point tracking, which is roughly the equivalent of driving a car for about a month without stopping, at full load.
The theoretical upper limit for an all-perovskite tandem is roughly 44%, which means there is still a lot of headroom. Current silicon solar panels top out commercially at around 22 to 24%, and the best laboratory silicon cells hit about 27%. At nearly 30%, the NIMTE tandem is already well ahead of what silicon can manage.
The catch, and it’s a significant one, is that perovskite solar cells still face real commercial hurdles that efficiency numbers alone don’t solve. Most high-efficiency perovskite devices use lead, which is a regulated substance in several major markets, and tin, which oxidizes readily and demands careful encapsulation. Manufacturing processes that work reliably at large scale, in ambient air rather than inert-atmosphere gloveboxes, are still being developed. Cost per watt remains an open question.
Still, the colloidal precursor strategy has something going for it that many previous approaches lack: it’s modular. The carboxylate anions are relatively cheap, the chemistry is in principle applicable to other perovskite compositions, and it doesn’t require redesigning the device architecture. If it holds up in follow-on work, it could be incorporated into existing fabrication processes without tearing everything down. That’s a rarer thing than it sounds in photovoltaics research, where efficiency gains are often purchased at the price of complexity that makes scale-up harder.
The broader competition shaping this work is worth keeping in mind. Perovskite-silicon tandem cells, where a perovskite top layer is deposited onto an existing silicon bottom cell, have already reached efficiencies above 33% in laboratory settings. All-perovskite tandems, where both layers are made of perovskite with no silicon at all, are cheaper in principle to manufacture but have lagged behind; pushing past 30% in a fully perovskite stack, reliably and at scale, is the milestone the field has been circling for several years. The NIMTE result doesn’t get there yet, but it narrows the gap, and it does so through a route that may prove more commercially tractable than some alternatives. What happens next probably depends less on the chemistry and more on whether manufacturers can be persuaded that the stability numbers hold up over years rather than months.
DOI / Source: https://doi.org/10.1016/j.joule.2025.102381
Discover more from SciChi
Subscribe to get the latest posts sent to your email.