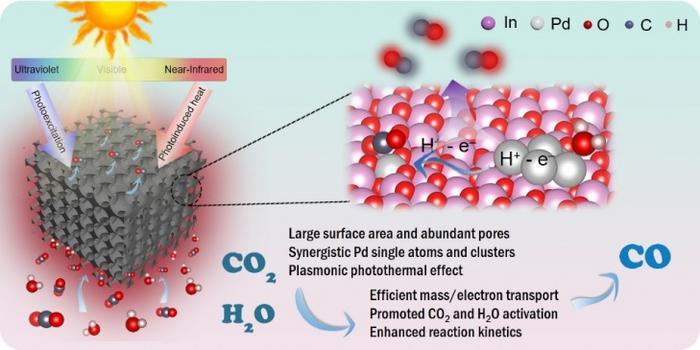
Light hits a particle of indium oxide, and something unusual happens. The palladium clusters dotting its surface don’t just absorb the photons. They convert them into heat, raising the catalyst’s skin temperature to around 230 degrees Celsius within seconds, even though the source of illumination is nothing more exotic than simulated sunlight. That warmth is not a side effect or a design flaw. It is, as a team at Zhejiang Sci-Tech University has now demonstrated, the whole point. The hotter the catalyst runs, the faster it splits CO2 and water into carbon monoxide, which can serve as a chemical feedstock or be processed further into liquid fuels.
The work, published in Chinese Journal of Catalysis, describes what the researchers call a photothermal catalyst, a material that exploits two mechanisms simultaneously rather than relying on either alone. The group, led by Prof. Benxia Li, designed the catalyst specifically to address a persistent problem in solar-driven CO2 conversion: that no single type of active site is actually good at all the jobs the reaction demands.
The challenge is deceptive in its simplicity. To make CO from CO2 and water using sunlight, a catalyst needs to do several things at once. It has to grab hold of CO2 molecules, which are chemically inert to the point of stubbornness. It has to split water to release the protons needed to drive the reaction forward. It has to shepherd electrons from one half-reaction to the other without losing them. And ideally, it ought to absorb light efficiently across as much of the solar spectrum as possible, not just the ultraviolet slice that most photocatalysts are tuned for. Designing a material that handles all of this in a coordinated way has proved difficult. Most research so far has treated each of these functions as separate engineering problems, optimizing them in isolation while the interactions between them remain poorly understood.
Yes, in principle, though the efficiency of photocatalytic systems is still well below what would be needed for commercial deployment. Researchers have demonstrated that carefully engineered catalyst materials can use sunlight to drive the reaction between CO2 and water, producing carbon monoxide, which can serve as a feedstock for synthetic fuels. The challenge is doing it fast enough, selectively enough, and with materials that don’t cost more to produce than the fuel they make.
CO2 is chemically very stable, meaning a lot of energy is needed to activate it. Light alone can supply that energy in the form of electrons, but the reaction also involves transferring protons from water, a step that proceeds much faster at elevated temperatures. By designing a catalyst that uses incoming light to generate its own heat through a physical process called plasmon resonance, the Zhejiang team found they could run both the photochemical and thermally accelerated parts of the reaction simultaneously in the same material.
Palladium has an unusual electronic structure that makes it effective at a range of catalytic tasks. Its outermost electrons sit energetically close to the level at which chemical reactions can proceed, allowing it to bind and activate CO2 molecules. Palladium clusters also interact strongly with visible light, making them efficient converters of photons into heat. Crucially, single palladium atoms and small clusters behave quite differently from each other, which is what allowed the researchers to assign each form a distinct role in the reaction sequence.
Broadly analogous, though the chemistry is quite different. Plants use sunlight to reduce CO2 using electrons ultimately derived from water, which is the same overall logic here. But plant photosynthesis operates through highly tuned protein machinery evolved over billions of years, at room temperature and with extraordinary efficiency at capturing light. Artificial photocatalysis is trying to achieve a similar input-output relationship using inorganic materials that are more robust and manufacturable, even if they can’t yet match biology’s elegance.
Li’s team took a different approach. Rather than picking a single type of active site, they built two kinds of palladium into the same support material, a porous indium oxide scaffold with a regular honeycomb-like architecture roughly a few hundred nanometers across. Some of the palladium sits as isolated single atoms, each anchored at a specific lattice site. The rest forms tiny clusters, just a handful of atoms apiece, scattered throughout the same structure.
The division of labor between them is quite specific. Single palladium atoms turn out to be the ideal sites for converting CO2 into CO. The electronic structure of palladium, with its d-band electrons close to the Fermi level, allows it to inject electrons into the lowest unoccupied orbital of CO2 molecules, activating them for further reaction. But isolated atoms struggle with the other half of the task: pulling hydrogen from water. That’s where the clusters come in. Palladium clusters are much more effective at dissociating water molecules, and the protons they liberate migrate across the catalyst surface to the single-atom sites where CO2 reduction is occurring. Quantum chemistry calculations support this picture, showing that clusters substantially lower the thermodynamic barrier to water dissociation.
There is a further wrinkle, and it turns out to be the most striking part of the story. Palladium clusters interact strongly with incoming light through a phenomenon called localized surface plasmon resonance. In simple terms, visible and near-infrared photons set the electrons in the clusters oscillating collectively, and when those oscillations decay, the energy dissipates as heat. This is the mechanism behind the temperature spike. Under the team’s experimental conditions, the catalyst surface runs hot enough to significantly accelerate reaction kinetics, essentially combining a photocatalyst and a thermal catalyst in a single material.
The results are not modest. The dual-site catalyst achieved a CO production rate nearly twenty times higher than plain indium oxide and about twelve times higher than an otherwise identical catalyst carrying only single-atom palladium, with no clusters. Selectivity for CO, rather than other products, came in at around 89 percent. The numbers matter because CO2 conversion catalysts are often criticized for producing mixed outputs that are difficult to use; high selectivity is what actually makes a material commercially relevant, not merely scientifically interesting.
The porous scaffold deserves credit too. Three-dimensional ordered macroporous architectures of this kind are sometimes described as photonic crystals, meaning the regular spacing of their internal pore walls scatters light in a way that increases photon residence time inside the material. More time in the material means more chances for absorption. The channels also improve mass transfer, allowing CO2 and water vapor to reach active sites more readily and reaction products to leave without getting trapped.
What the paper doesn’t yet address is scalability. Synthesizing the material involves template-assisted pyrolysis using polystyrene microspheres, followed by careful thermal treatment in a hydrogen-argon atmosphere to coax the palladium into its dual atomic configuration. That is manageable at laboratory scale, but the process adds complexity. Whether the approach translates to gram-scale or kilogram-scale production without the catalyst’s structure degrading or the single-atom and cluster ratio shifting is an open question. Palladium is also not cheap, and photocatalytic systems still lag well behind electrocatalytic ones in terms of overall solar-to-fuel efficiency.
Still, the conceptual advance here seems genuinely useful. The idea that a single catalyst can harvest light, convert part of that energy directly into useful heat, and deploy two chemically distinct active sites in coordinated sequence addresses something that piecemeal approaches cannot. As the researchers note, most previous studies tuned the CO2 reduction and water oxidation half-reactions separately, without accounting for how changes in one alter the kinetics of the other. Treating them as an integrated system, rather than independent modules, produced a catalyst that outperformed the parts.
For a field that tends to measure progress in increments, that is a meaningful shift in framing. It suggests the bottleneck in solar CO2 conversion may be less about finding better materials in isolation than about designing systems where different functional components actually talk to each other. Whether palladium-loaded indium oxide becomes a practical technology or a proof-of-concept footnote depends on what the chemistry does at industrial operating conditions. But the logic of the approach, using heat the catalyst generates itself to close a kinetic gap its light-driven chemistry can’t quite bridge, is the kind of idea that tends to survive contact with messier reality.
DOI: 10.1016/S1872-2067(25)64919-9
Discover more from SciChi
Subscribe to get the latest posts sent to your email.