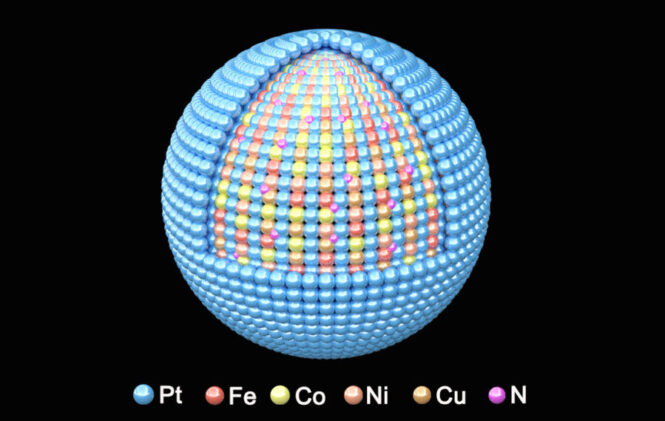
A new recipe mixing five metals in precisely controlled atomic arrangements has delivered what heavy duty vehicle manufacturers have been waiting for: a fuel cell catalyst tough enough to handle the grueling demands of long haul trucking while maintaining the efficiency needed for commercial viability.
The breakthrough comes from researchers at Brookhaven National Laboratory, who developed what they call a “high entropy intermetallic” catalyst by combining platinum, iron, cobalt, nickel, and copper with nitrogen in a carefully orchestrated atomic structure. Their work, published in Nature Communications, addresses one of the most stubborn challenges in fuel cell technology, creating catalysts that can withstand the harsh conditions inside fuel cells powering trucks, buses, and other heavy duty vehicles.
Beyond Passenger Cars
While fuel cells have found success in passenger vehicles, the heavy duty market presents different challenges entirely. Trucks need to run continuously for thousands of hours under high heat and acidic conditions that would quickly degrade conventional catalysts. The research team, led by Kotaro Sasaki and Xueru Zhao from Brookhaven’s Chemistry Division, set out to design materials that could handle these extreme environments.
“Catalysts are the components that enable the chemistry at the electrodes inside a fuel cell. These materials, often made of metals, bring the reacting chemicals together and lower the energy required to run the reaction. But the catalyst has to be able to perform this function over and over in challenging conditions, such as high heat or a harsh acidic environment.”
The team’s approach centered on exploiting what materials scientists call “high entropy” effects: the stabilizing influence that emerges when multiple elements are arranged in ordered atomic structures. By incorporating five different metals rather than the typical two or three, they created a catalyst with unusual durability characteristics.
Using advanced imaging techniques at Brookhaven’s National Synchrotron Light Source II, the researchers discovered something unexpected in their new material’s atomic structure. The atoms weren’t sitting exactly where theory predicted they should be. Instead, they found tiny distortions measuring much less than the width of a single atom – what they termed “sub-angstrom strain.”
Meeting Industry Demands
These microscopic imperfections, combined with strong bonds between the metals and nitrogen atoms, turned out to be the catalyst’s secret weapon. In laboratory tests designed to simulate the operating conditions of heavy duty trucks, the new catalyst maintained excellent performance over 90,000 operating cycles, equivalent to continuous operation for 25,000 hours.
The numbers tell the story of a material that could finally make hydrogen fuel cells viable for commercial trucking fleets. After those 90,000 cycles, the catalyst was still producing current densities well above Department of Energy targets for heavy duty vehicle applications. In real world terms, this translates to fuel cells that could power trucks for hundreds of thousands of miles without significant performance degradation.
“These results show a practical pathway to building fuel-cell systems that can power the trucks and buses of tomorrow. Our catalyst not only meets immediate market needs but also lays the groundwork for widespread adoption in heavy-duty transportation.”
The catalyst’s performance in membrane electrode assembly tests, closer to real fuel cell conditions, was equally impressive. Under conditions mimicking heavy duty vehicle operation, it achieved current densities of 1,388 milliamps per square centimeter after 90,000 cycles, surpassing the Department of Energy’s Million Mile Fuel Cell Truck consortium target of 1,300 milliamps per square centimeter.
The research represents a convergence of multiple scientific disciplines within Brookhaven Lab. Teams from surface electrochemistry, materials science, and advanced characterization facilities collaborated to understand not just what makes the catalyst work, but why it works at the atomic level. This fundamental understanding could accelerate the development of even more advanced catalysts for fuel cell applications.
What makes this development particularly significant is its timing. As transportation companies face increasing pressure to reduce emissions, hydrogen fuel cells offer one of the few viable pathways to zero emission heavy duty vehicles. Unlike battery electric trucks, which face range and charging time limitations for long haul applications, fuel cell trucks can be refueled quickly and offer the range needed for commercial freight operations.
The work was supported by the Department of Energy as part of broader efforts to advance fuel cell technologies for heavy-duty vehicle commercialization. The research demonstrates how fundamental scientific discoveries at national laboratories can translate into practical technologies that address real-world industrial challenges.
For an industry that has long struggled with the durability demands of heavy-duty applications, this catalyst represents more than just an incremental improvement. It offers a potential solution to one of the key technical barriers preventing widespread adoption of fuel cell technology in commercial transportation – a market that could significantly impact efforts to decarbonize the freight industry.
Nature Communications: 10.1038/s41467-024-54175-0
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.