Fibroblasts do not normally care what shape they make. Put a few million of them on a soft gel in a Barcelona lab, wait three days, and they will crawl into long aligned bands like iron filings over a magnet, content to stretch themselves out and pull mildly on whatever they are sitting on. What they will not usually do is decide, all at once, to curl into a bowl. But that is the trick a team at the Institute for Bioengineering of Catalonia has now pulled off, using nothing more exotic than a chemical stencil and a dose of enzyme.
Their report, published today in Science, describes the first method for designing the three-dimensional shape a living tissue will adopt before the tissue has adopted it. The researchers call the result a shape-programmable living surface, which is a mouthful, though what it actually looks like on video is rather homelier: a flat disc of cells that suddenly, over the course of a few minutes, reaches up and folds itself into a cup, or a saddle, or something uncannily reminiscent of a rose.
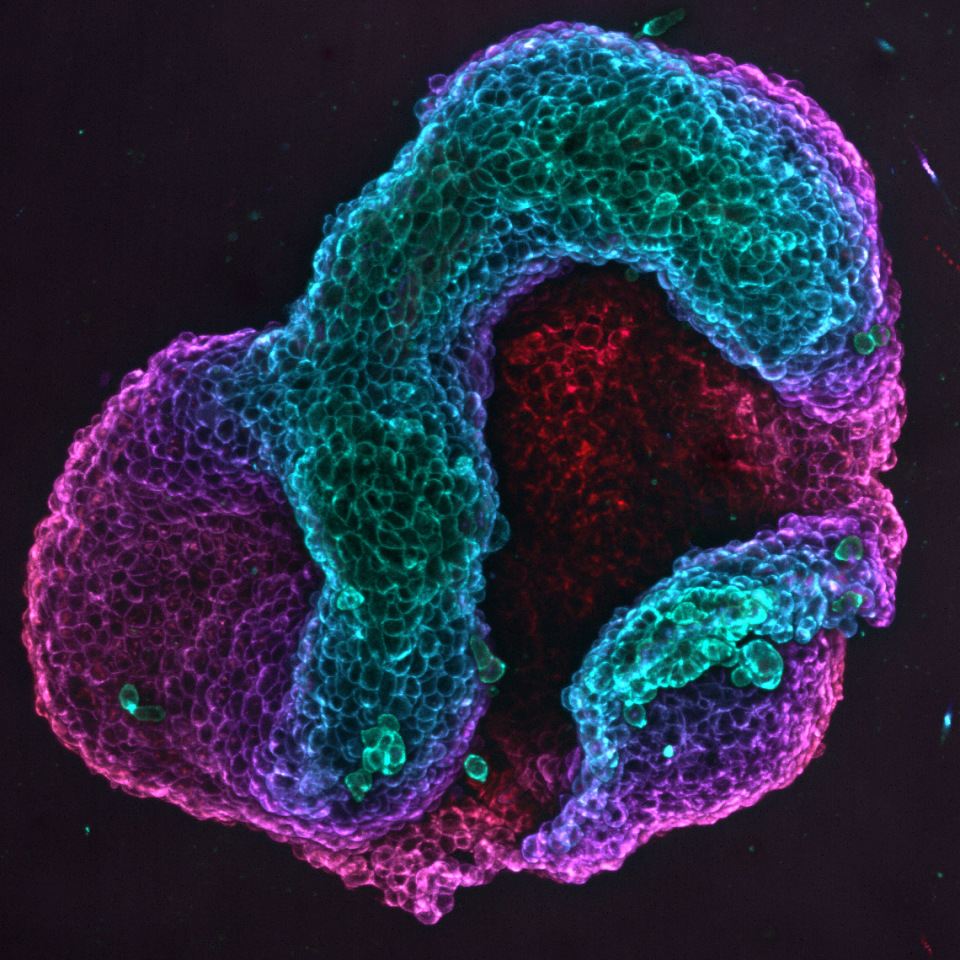
The defects doing the work
The physics underneath is borrowed, slightly unexpectedly, from liquid crystals. Elongated cells, like the rodlike molecules in an LCD screen, tend to line up with their neighbors, creating what physicists call nematic order. It is the reason a monolayer of fibroblasts, seen under phase-contrast, looks faintly like the grain of a piece of wood. Occasionally the alignment breaks down at a point, producing a little whorl or bifurcation that mathematicians call a topological defect. These defects are the same sort of thing you see at the centre of a fingerprint, and cells, it turns out, pull hardest around them.
That much had been known for years. The open question was whether you could boss the defects around.
To do so, Pau Guillamat, the paper’s first author, and his colleagues built a kind of invisible railway for the cells. They patterned soft gels with fibronectin, a sticky matrix protein, in thin adhesive lines about two micrometres across, and surrounded those lines with a cell-repellent polymer. The fibroblasts, dropped onto the gel, crawled onto the adhesive stripes, aligned along them, and by the third day had produced a tissue in which the topological defects sat exactly where the pattern said they should. “the forces can control the generation of a three-dimensional shape,” says Guillamat, which is the payoff line but also, importantly, a testable claim.
Peeling the skin
The test went like this. While the tissue was still glued to the substrate below it, the stresses the cells generated had nowhere to go; they were anchored in place, the way a stretched sheet of cling film stays flat if you keep your hands on the corners. To release those stresses, the team dissolved the extracellular matrix with collagenase, the same enzyme meat tenderisers use. Within five to ten minutes, the tissue peeled off. Within a few more, it began to fold.
The shapes that emerged were not random. A circular disc containing two topological defects, arranged at opposite ends, consistently collapsed into a bowl. Four defects gave a structure with threefold symmetry. Six defects produced something fourfold. Move the defects closer together and the bowl got deeper; rotate the pair by forty-five degrees and the whole folding axis rotated with it. In one configuration, where the team engineered a mix of compressive and tensile zones, the tissue came up with two bowl-shaped invaginations joined by a saddle, a morphology you would be hard-pressed to design on purpose with any conventional scaffold.
To understand why, Marino Arroyo’s group at the Polytechnic University of Catalonia and CIMNE built a theoretical model treating the cell sheet as a contractile elastic shell with a nematic texture baked into it. When the active stresses in such a sheet cannot relax by shrinking in the plane, the shell buckles out of it; this is the same principle, though in a rather different guise, that causes a wet lily leaf to curl or a damaged pea pod to twist. Botanists call it Gaussian morphing, a consequence of Gauss’s 19th-century Theorema Egregium, which says that a flat sheet cannot be smoothly mapped onto a curved one without stretching somewhere. The cells, in other words, are doing what plants do, not what animal embryos usually do. Animal tissues generally fold through active bending or compressive buckling; this is something else.
It should be said, plainly, that these are fibroblasts in a dish, not organs. The team used blebbistatin, a drug that weakens cellular contractility, to stop the tissues tearing themselves apart during retraction; without it, the stresses are fierce enough to fracture the sheet.
And the morphologies, although stable for hours, slowly keep contracting. The structures are also, for now, rather simple. A bowl is not a kidney.
Where this goes
Still, the implications are not modest. If you can decide in advance the geometry a tissue will adopt, you gain a new route into tissue engineering that does not depend on printing a scaffold and hoping the cells comply. Biohybrid robotics, in which pieces of living muscle serve as actuators, suddenly has a way to build curved parts rather than just flat ones. And the same toolkit allows developmental biologists to ask questions that were previously fiddly to pose at all, such as what a tumour does when you hand it a pattern of stresses it did not generate itself. Xavier Trepat, who co-led the study with Arroyo, calls the system “a perfect tool for understanding how patterns of cell orientation influence the mechanics” of complex tissues, and given how little access biologists have had to this layer of morphogenetic control, the description is not overblown.
What the Barcelona group has really shown is that the information needed to build a three-dimensional shape can be smuggled in two dimensions, written down as a pattern of stripes a child could draw. The tissue reads it, and folds.
Frequently Asked Questions
How do the cells know what shape to make?
They don’t, not in any cognitive sense. The researchers pre-pattern a gel with sticky stripes that force the cells to line up in a chosen direction, which creates predictable stress fields across the sheet. When the tissue is peeled free, those stresses have no option but to relax by buckling out of the plane, and the geometry of the buckle is dictated entirely by where the alignment pattern placed the topological defects.
Is this the same as 3D bioprinting?
No, and that is the point. Bioprinting builds structure by depositing cells onto or within a scaffold in the shape you want. This method starts with a flat sheet and lets the cells generate their own three-dimensional form through internal mechanical forces, which means no external scaffold is required. It is closer, conceptually, to how a flat sheet of paper becomes an origami crane than to how a 3D printer works.
Could this be used to grow organs?
Not yet, and not soon. The shapes demonstrated so far are relatively simple geometries like bowls and saddles made from a single cell type, whereas organs involve many cell types in complex hierarchical arrangements. What the method does offer is a new layer of control that could, in principle, be combined with existing organoid techniques to impose specific geometries during development. That is a long way from a functional transplant, but it is a piece of the puzzle that was previously missing.
Why does the tissue fold like a plant rather than like an embryo?
Animal embryos usually fold through active bending or by buckling under compression, mechanisms that involve cells actively pushing or squeezing in coordinated ways. The Barcelona tissues fold for a different reason: their internal tensions are geometrically incompatible with staying flat, a phenomenon called Gaussian morphing that plant biologists use to explain how leaves curl and flowers open. The researchers essentially demonstrated that animal cells can be coaxed into using a morphing strategy normally seen in botany.
What could biohybrid robots actually do with this?
Current biohybrid robots use strips of muscle tissue to pull on simple skeletons, which limits their motion to bending and contraction. If curved, programmable living surfaces can be built reliably, a robot could in principle include components that fold, cup or grip, which are motions that are awkward to achieve with flat actuators. The more immediate payoff is probably in research tools rather than walking machines, but the direction of travel is clear.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.

