There’s a mineral called bustamentite that almost nobody studies. It forms in flat hexagonal plates, grows readily in warm water, and turns up as an unwanted contaminant in perovskite solar cells. For decades it sat at the margins of materials science, useful mostly as a precursor for more fashionable compounds. Then a group of physicists decided to look at it very carefully in the terahertz range, and found something that had been hiding in plain sight since the 1970s: bustamentite, better known as lead iodide, can compress light to a degree that has no real parallel among materials tested at these frequencies.
The finding, published in Nature Communications, positions lead iodide as the first material to bring the strange physics of hyperbolic phonon-polaritons into the deep terahertz, a spectral region that the nanophotonics community has been trying to crack for years.
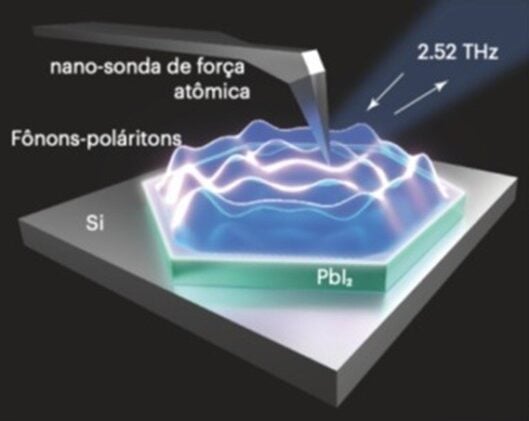
Phonon-polaritons are hybrid quasiparticles. In polar crystals like lead iodide, light and atomic vibrations can couple so tightly that neither propagates independently, and what you get instead is something with properties neither possesses alone. “It’s as if the phonon were dressed in light,” says Raul de Oliveira Freitas, who coordinates the Imbuia beamline at Brazil’s synchrotron facility LNLS-CNPEM and led the study. “The propagation characteristics and interaction with matter of these quasiparticles differ from both isolated light and isolated phonons.” In materials with a particular kind of crystalline anisotropy, these hybrids become hyperbolic: they travel through the bulk of the crystal along cone-shaped wavefronts, guided by the geometry of the lattice itself rather than by conventional optical boundaries.
Why Terahertz Is Hard
The terahertz gap has frustrated physicists partly because of scale. Light in this range has wavelengths of hundreds of micrometers. That’s a problem if you want to route it through circuits that fit on a chip. Confining terahertz radiation to the nanoscale requires suppressing the diffraction limit, the fundamental constraint that prevents ordinary optics from resolving or manipulating anything smaller than roughly half the wavelength. “In classical optics, it isn’t possible to observe or manipulate structures much smaller than the wavelength of light,” Freitas says. “With polaritons, we’ve managed to overcome that limit.”
Several materials had already shown this trick in the infrared range. Hexagonal boron nitride is the canonical example, with polaritons that propagate with low losses and squeeze infrared wavelengths into extraordinarily small volumes. But pushing comparable behavior to longer terahertz wavelengths meant finding a material with the right anisotropy at the right frequencies, and most of the obvious candidates didn’t have it. Lead iodide, it turns out, does. Its crystal structure produces a pronounced mismatch in how it responds to electric fields along different axes, generating a hyperbolic response in a band that runs from about 1.55 to 3.03 terahertz, a range that overlaps neatly with where next-generation wireless communication systems are heading. “Today, Wi-Fi and 5G operate at frequencies of a few gigahertz,” Freitas notes. “But there is interest in moving toward hundreds of gigahertz, or even terahertz, because the higher the frequency, the greater the bandwidth and data transmission capacity.”
To see the polaritons directly, the team used scattering-type near-field optical microscopy, a technique in which a platinum-coated atomic force microscope tip is illuminated by a terahertz laser. The tip acts as an antenna, concentrating the field at its apex to a hotspot of a few tens of nanometers. “The electric field density in s-SNOM probes is up to 105 times higher than in free waves,” Freitas says. The result is that a wave 200 micrometers long gets squeezed into a volume smaller than 50 nanometers. In the images from the microscope, the polaritons appear as ripple patterns of alternating bright and dark fringes, propagating outward from the edges of thin lead iodide crystals with a regularity that matched theory almost exactly.
A Quality Factor That Surprises
Quality factor is a measure of how long a system sustains oscillation before dissipating its energy as heat. High quality factors are what separate a useful resonator from a leaky one. Lead iodide’s quality factor in the terahertz range, assessed by the team using a figure of merit that tracks how many oscillation cycles a polariton completes before it damps out, reached 17 for a crystal 340 nanometers thick. That’s on par with hexagonal boron nitride in its native infrared range, and comfortably above molybdenum trioxide, the other leading benchmark material, whose maximum in the mid-infrared is around 12. “The longer the system oscillates, the higher the quality factor,” Freitas says. “PbI2 performed comparably to hexagonal boron nitride, which is the reference material in the infrared range.”
The confinement numbers are perhaps more striking. A 144-nanometer-thick flake compressed the terahertz wavelength by a factor of 264: that is, the polariton wavelength inside the crystal was 264 times shorter than the free-space wavelength of the light driving it. Go to a flake thinner than 100 nanometers and the factor exceeds 300. This happens because thinner crystals force the polariton into a tighter mode, and lead iodide’s high ionicity, a large charge transfer in its chemical bonds, sustains the coupling without extracting too heavy a penalty in losses. The material’s extreme dielectric anisotropy, captured in a Lyddane-Sachs-Teller ratio of 4.20 (versus 1.41 for hexagonal boron nitride), gives it a hyperbolic band that is far broader than in comparable materials, which translates into more usable frequencies and more flexible device design.
What makes the finding practically appealing is how unremarkable lead iodide is to produce. Growing hexagonal boron nitride to research quality requires extreme pressure, high temperatures, and decades of accumulated expertise; only a handful of groups worldwide can do it reliably. Lead iodide crystallizes from water. “Simply dissolve the salt in water until a supersaturated solution is obtained and heat it to about 80 degrees Celsius,” Freitas explains, “something that can be done on a household stove. During cooling, the material crystallizes, forming structures that can be collected.” The synthesis is hydrothermal, the precursors are cheap and common, and because iodine is monoisotopic and lead has minimal natural isotopic variation, the resulting crystals are reproducible in a way that matters enormously for device fabrication.
The broader vision is for photonic circuits inside chips, where information moves on light rather than electrons. “Currently, information is transmitted within devices via electrons,” Freitas says. “Using light can drastically increase speed and reduce losses. It’s analogous to what happened in the field of telecommunications. Before, we used electrical cables; today, we use optical fibers.” The same logic, applied at the scale of integrated circuits, suggests a future where processor-to-processor communication happens at the speed of light through terahertz waveguides, consuming a fraction of the energy that copper interconnects require. The team has designed experiments around lead iodide functioning as a resonator, a beam splitter, and a modulator; all three functions are demonstrated in the paper.
The material’s role in perovskite research adds an adjacent strand to the story. Lead iodide is a ubiquitous precursor for the perovskite compounds now competing seriously with silicon in solar cell efficiency tables, and understanding its optical phonon behavior may help explain why excess lead iodide in perovskite films sometimes passivates defects and sometimes accelerates degradation. Whether or not that turns out to be a useful connection, “the expectation of the scientific community,” Freitas says, “is to make light circuits increasingly present in everyday devices.” The material that was cluttering up solar cells may turn out to be what makes that possible.
DOI: 10.1038/s41467-026-69027-6
Frequently Asked Questions
Why does compressing terahertz light to the nanoscale matter for future technology?
Terahertz radiation has wavelengths of hundreds of micrometers in free space, which makes it impossible to route through chip-scale components using conventional optics. Phonon-polaritons in lead iodide compress that wavelength by a factor of 264 or more, shrinking the effective size of the light to a point where it could feasibly travel through nanoscale waveguides and resonators on a chip. That opens a path to optical data links inside devices that would be dramatically faster and more energy-efficient than the copper interconnects used today.
How is lead iodide different from the materials researchers normally use for this kind of physics?
Hexagonal boron nitride is the gold standard for phonon-polariton research, but it only works in the infrared range and is notoriously difficult to synthesize at research quality. Lead iodide crystallizes from warm water in a process that requires no specialist equipment, and its natural isotopic consistency means crystals are reproducible. Crucially, it operates in the terahertz range rather than the infrared, which is where next-generation wireless communication is heading.
Is lead iodide safe to work with at this scale?
Lead compounds require standard laboratory precautions because of lead’s well-documented toxicity. The paper does not address this directly, but lead iodide is already handled routinely in perovskite solar cell research, where it is synthesized and processed in academic and industrial labs worldwide. Whether its toxicity profile presents a barrier to large-scale chip integration is a practical question that research at this stage has not yet confronted.
Could this technology actually replace electronic circuits in the near future?
The research is at the basic science stage, demonstrating that the material can confine and guide terahertz light with low losses. The gap between a proof-of-concept polariton waveguide and a manufacturable photonic chip is significant, involving challenges in fabrication, integration with existing electronics, and device engineering that remain largely unsolved. The researchers describe lead iodide as a candidate for resonators, beam splitters, and modulators, which are the building blocks for circuits, but none of those components have been built into working devices yet.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.