A single calf muscle contains hundreds of tiny nerve-to-muscle connections, each one firing in precise sequence to produce something as mundane as standing still. Motor units, they’re called. Think of them as individual musicians in an orchestra, each responding to a shared conductor’s cue so the whole ensemble sounds smooth rather than chaotic. For most of us this coordination happens without a flicker of conscious thought. But after an incomplete spinal cord injury, even when a person can walk again, that internal orchestra may be playing from a scrambled score.
Key Takeaways
- The study reveals that people with incomplete spinal cord injuries struggle with balance due to less refined nerve signals coordinating muscle units.
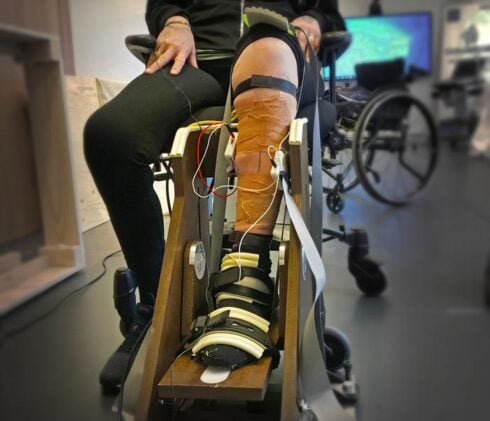
- Researchers used high-density electromyography to examine motor unit behavior in the calf muscles of individuals with and without spinal cord injuries.
- At low effort, SCI individuals showed less synchronization among motor units, leading to shaky and unstable movements.
- At higher effort, SCI patients displayed stronger but less precise low-frequency synchronization, pointing to a compensatory strategy of the nervous system.
- These findings may lead to new rehabilitation approaches that focus on neural coordination, rather than just functional movement.
Researchers at KTH Royal Institute of Technology in Stockholm have, for the first time, examined how these motor units work together (or fail to) in people living with incomplete spinal cord injuries. Their findings, published in the Journal of NeuroEngineering and Rehabilitation, suggest the nervous system’s strategy for controlling movement shifts in ways that clinical assessments typically miss.
The study focused on two muscles in the calf, the soleus and the gastrocnemius medialis, both of which work in tandem during everyday tasks like standing, walking, and pushing off a step. The team used high-density electromyography, a grid of surface sensors that picks up electrical chatter from individual motor units through the skin. Twenty-five volunteers (fifteen with incomplete spinal cord injuries and 10 without) pushed against a force-measuring device at two levels of effort: a gentle 20 percent of their maximum, and a moderate 50 percent. No needles, no invasive procedures. Ruoli Wang, associate professor in biomechanics at KTH’s Promobilia MoveAbility Lab, says the researchers’ approach was completely non-invasive.
What emerged was, in a way, two different problems at two different volumes.
At the lower effort level, the motor units in the SCI group were less likely to belong to what the researchers call the “shared cluster,” a population of nerve-muscle connections driven by the same coordinated signal from the brain and spinal cord. Lead author Zhihao Duan, a PhD student at KTH, puts it plainly. “They were much less being driven by the same coordinated signal from the nervous system,” he says. The practical upshot? Shaky, unstable movements during tasks that should feel effortless. Standing quietly. Holding a coffee cup steady. That sort of thing.
Crank the effort up to 50 percent, though, and a different pattern shows up. The SCI group displayed stronger low-frequency synchronization between the two calf muscles, specifically in what neuroscientists call the delta band (below 5 hertz). Because those low-frequency signals are what actually translate into muscle force, this heightened synchrony could mean the nervous system is, essentially, shouting. Duan suspects the nervous system is compensating by sending louder, less refined signals.
It is perhaps the contrast between these two effort levels that tells the most revealing story. In people without injuries, the nervous system smoothly recalibrates its strategy as demands increase, shifting which clusters of motor units it leans on. The SCI group showed no such flexibility. Their cluster distribution stayed roughly the same whether they were pushing gently or moderately. Wang describes it as a kind of neural rigidity. “One interesting finding is that after spinal cord injury the nervous system becomes more rigid and less able to change its approach as the muscles work harder,” she says. “A healthy nervous system on the other hand is able to adapt its strategy as force demands, to adjust the shared neural drive level.”
The underlying mechanics here are worth unpacking, even if they get a bit technical. Motor units don’t operate independently; they receive two kinds of input from the central nervous system. There’s the common input, shared across a pool of neurons and responsible for coordinated activation, and then independent input unique to each neuron. The balance between these determines how smoothly (or not) a muscle produces force. Using factor analysis on the decomposed spike trains, the researchers could sort motor units into clusters based on which inputs they shared. Three groups emerged: units loyal to their own muscle, units loyal to the partner muscle, and units receiving shared input from both. It was that shared cluster, the glue holding the two muscles’ coordination together, that shrank in people with SCI at low effort levels.
The sample was small, roughly two dozen participants, and the team acknowledges that surface-based decomposition can only capture a fraction of the motor units actually active in any given contraction. The SCI group was also heterogeneous: injury levels ranged from cervical to lumbar, with time since injury spanning from months to over a decade. All of which means the findings need replication in larger, more carefully stratified cohorts.
Still, the results could point toward something clinically useful. Right now, rehabilitation after spinal cord injury relies heavily on functional assessments: can the person walk, how far, how fast. These measures miss what’s happening at the level of neural coordination. A person might walk across a room and look fine, but their nervous system could be working overtime, compensating with blunt-force synchrony rather than the refined, flexible control a healthy system uses.
“This finding may open the door to a new rehabilitation biomarker, helping clinicians and researchers design new neurorehabilitation strategies to re-tune the spinal cord control and to restore coordinated neural input,” Wang says. The idea is appealing, if still speculative. If clinicians could track motor unit synergies over time, they might be able to tailor rehabilitation programs not just to what a patient can do, but to how their nervous system is actually doing it.
Whether that kind of precision monitoring becomes practical depends on the technology catching up. HD-EMG grids are research tools for now, not something you’d find in a typical rehab clinic. But the cost of surface sensor arrays has been dropping for years, and the decomposition algorithms that pull individual motor unit signals out of the noise keep getting sharper. It is not hard to imagine a future where a set of sticky electrode patches on someone’s calf could reveal what no amount of walking tests ever could.
DOI: 10.1186/s12984-026-01874-2
Walking is a relatively coarse motor task compared to standing still or producing a smooth, controlled force. After an incomplete spinal cord injury, the nerve signals that coordinate individual motor units inside muscles become less refined, even if enough gross function remains for locomotion. The result is that someone can get from A to B on foot but may wobble when standing quietly or find it difficult to hold a steady grip, because the fine-grained neural coordination behind those tasks has been disrupted at a level that standard clinical walking tests simply do not measure.
They used high-density electromyography, which involves placing a grid of small surface electrodes on the skin over the calf muscles. Advanced decomposition algorithms then tease apart the overlapping electrical signals to identify the firing patterns of individual motor units underneath. The technique has been around in research settings for some years, but this is the first time it has been applied to study motor unit synergies specifically in people with spinal cord injuries.
Not yet, but the groundwork is there. The sensor arrays and software used in this study are still research-grade tools, and the sample sizes are too small to establish clinical norms. However, if the approach is validated in larger trials, it could eventually give clinicians a way to track how a patient’s neural coordination is recovering, not just whether they can perform a given movement, which would allow rehabilitation programs to be tailored at a much deeper level.
At higher levels of muscle effort, the researchers found that the injured nervous system shows stronger low-frequency synchronization between the two calf muscles studied. Think of it as the brain turning up the volume on a single, blunt command rather than issuing nuanced instructions to different groups of motor units. This compensatory strategy helps generate the needed force, but it sacrifices precision, which could explain why people with spinal cord injuries often find moderate-effort tasks feel less controlled or more fatiguing than they should.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.