Cavity villains may owe their power to two tiny sticky molecules. In new work from UC Berkeley, researchers mapped gene clusters in the oral microbiome and found a pair of metabolites that help bacteria grip teeth and each other.
Published August 19, 2025 in Proceedings of the National Academy of Sciences, the study ties a specific biosynthetic gene cluster to robust plaque formation, including in strains of Streptococcus mutans, the classic culprit in tooth decay.
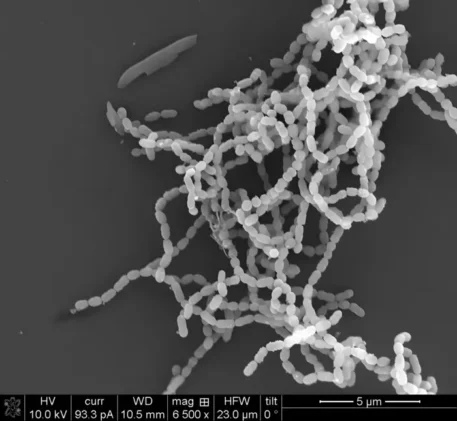
The twist is chemistry. The cluster makes two small lipopeptides, nicknamed mutanoclumpins, that act like social glue. One favors clumps. The other favors strings. Together they turn loose cells into a fortress of biofilm that resists a rinse and a prayer. This is not just a S. mutans story either. The same genetic cassette shows up in multiple oral species, hinting at a broader playbook for life on enamel.
The team did not chase species. They chased genes. Scanning mouth metagenomes for biosynthetic clusters linked to disease, they flagged one hybrid NRPS-PKS pathway that correlated with caries across independent datasets, then purified its products and watched what they do. In knockout experiments, S. mutans missing the cluster struggled to form a durable biofilm. Add back both purified mutanoclumpins at physiologic levels and the strong biofilm snapped right back. Add just one, and the magic fizzled.
“Particular strains belonging to the same species can be a pathogen or a commensal or even probiotic,” Zhang said.
That line lands harder than it first appears. If pathogenic punch lives in accessory gene cassettes and the molecules they make, then species labels are a blunt instrument. The practical upshot is strangely hopeful: instead of carpet-bombing plaque, we might retool it. The researchers suggest giving favored commensals a competitive boost, for example by equipping Streptococcus salivarius, a marketed oral probiotic, with better sticking power so it can hold territory against acid producers.
There are caveats. Stronger biofilms are not automatically good, and the same sticky chemistry that helps friendly neighbors hold ground can also harden the defenses of the usual suspects. The study nudges toward dual strategies, inhibition and engineering. If mutanoclumpins are truly key to cariogenic architecture, targeted inhibitors could soften the scaffold so brushing and saliva shear can do their work. On the flip side, a probiotic could be tuned to colonize first and crowd out acid factories. The dental aisle might get more interesting, and yes, commercial stakes are embedded here.
The science also delivers a rare mechanistic flourish. Mutanoclumpins come in two major flavors: a macrocycle, MC-584, and a linear cousin, MC-586. The macrocycle binds tightly in the cell membrane and drives three-dimensional clumps. The linear molecule binds cell wall and membrane and encourages chain-like strings. Clumps and strings. Repeat that, because it matters. The biofilm’s toughness depends on both, a cooperative mode of action that is uncommon for natural product pathways. Disrupt either motif and the architecture loses integrity.
“Our future work will be to create a broad map of the collection of these specialized metabolites to look at collectively what this dynamic, complex community on your teeth is making,” Zhang said.
A buried lede here: the same gene-level approach previously turned up an antibiotic made by oral bacteria. In other words, the mouth is not just a battlefield, it is a chemical factory hiding levers we have barely touched. Turns out, the plaque we curse every morning carries its own drug discovery pipeline. That does not mean we can toss our toothbrushes. As first author McKenna Yao reminds us elsewhere, brushing still removes biofilm best. But the real surprise came when two molecules, acting together, proved sufficient to restore a rugged biofilm phenotype in a crippled strain.
So, can good bacteria become probiotic cavity fighters? Maybe. The path runs through sticky chemistry, not species checklists. If we can dial down clumps and strings in the wrong mouths and dial them up in the right ones, we might shift the balance from dissolution to defense. It is an elegant idea, and like most elegant ideas in biology, it will meet the mess of real mouths. That is fine. Biology is messy. Teeth are, too.
Bacteria on teeth build biofilms, the sticky communities we call plaque. This study links a specific gene cluster to two small molecules, mutanoclumpins, that help cells stick and organize. One molecule promotes tight clumps, the other promotes chain-like strings. Together, they create a tougher biofilm. When researchers deleted the cluster, biofilms weakened. Adding both purified molecules restored strength. Because this chemistry sits in a mobile genetic cassette, it could be engineered. Two future routes follow: design inhibitors that block mutanoclumpins to weaken harmful biofilms, or boost beneficial species with better adhesion so they outcompete acid-producing microbes that drive cavities.
Journal: Proceedings of the National Academy of Sciences
DOI: 10.1073/pnas.2504492122
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.