Water trapped in spaces smaller than a virus has revealed a paradoxical new phase that challenges our understanding of the world’s most essential substance. Japanese scientists have discovered that confined water can exist simultaneously as both frozen and liquid-like, creating what they term a “premelting state” that defies conventional physics.
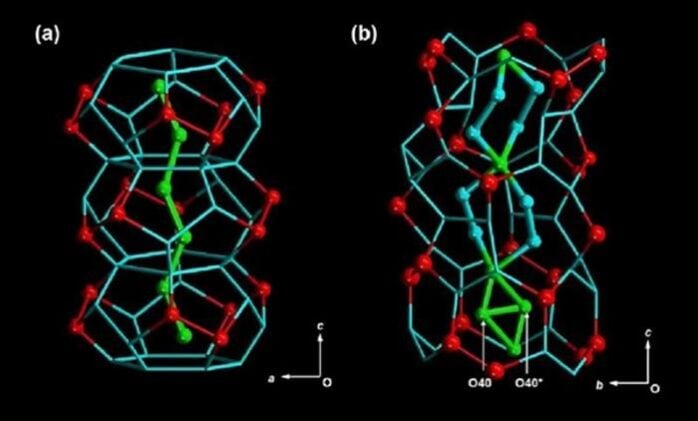
The discovery emerged from painstaking analysis of water molecules squeezed into crystalline channels just 1.6 nanometers wide—spaces so narrow that only a few dozen water molecules can fit across. Using nuclear magnetic resonance spectroscopy, researchers at Tokyo University of Science found that these confined water clusters organize themselves into a three-layered hierarchy with dramatically different behaviors from ordinary water.
“The premelting state involves the melting of incompletely hydrogen-bonded H2O before the completely frozen ice structure starts melting during the heating process. It essentially constitutes a novel phase of water in which frozen H2O layers and slowly moving H2O coexist.”
Professor Makoto Tadokoro, who led the research team, explains that this strange state occurs because water molecules near the crystal walls behave differently than those floating in the center of the nanochannels. The confined environment creates a gradient of molecular behavior that doesn’t exist in bulk water.
A Molecular Hierarchy Emerges
The research, published in the Journal of the American Chemical Society, reveals that confined water naturally arranges itself into three distinct layers. Water molecules in the primary layer bond directly to the crystal walls and move relatively slowly. The secondary layer contains molecules that interact with the primary layer but maintain more freedom. At the center, in what researchers call the tertiary region, water molecules spin rapidly and resist freezing altogether.
This hierarchical structure becomes particularly intriguing during temperature changes. As the confined water warms up, it doesn’t simply transition from solid to liquid like normal ice. Instead, it passes through the newly identified premelting phase where some molecules remain frozen while others begin moving with liquid-like mobility.
The measurements revealed correlation times remarkably similar to liquid water, even though the molecules’ positions remained relatively fixed like a solid. This contradiction suggests that while water molecules couldn’t easily change their locations, they could rotate and vibrate with surprising freedom.
Implications Beyond Basic Science
The discovery has implications far beyond academic curiosity. Confined water plays crucial roles in biological processes, from protein function to cellular transport. Understanding how water behaves in tight spaces could illuminate how life operates at the molecular level.
“By creating new ice network structures, it may be possible to store energetic gases such as hydrogen and methane and develop water-based materials such as artificial gas hydrates.”
The research also opens possibilities for technological applications. The ability to control water’s freezing properties based on confinement could lead to new materials for energy storage, particularly for capturing and storing gases like hydrogen and methane in stable, water-based structures.
The team’s approach combined multiple analytical techniques to overcome longstanding limitations in studying confined water. Traditional X-ray analysis struggles to track hydrogen atoms, which are crucial for understanding water’s behavior. Nuclear magnetic resonance spectroscopy provided the sensitivity needed to observe individual water molecules rotating and moving on picosecond timescales.
Their crystals, grown over three weeks through careful chemical diffusion, created perfect test chambers for water confinement studies. The hexagonal rod-like crystals contained parallel channels that acted like molecular-scale test tubes, allowing researchers to observe water behavior under precise conditions.
The discovery adds another layer to water’s already complex personality. Scientists have identified at least 20 different forms of ice, each with unique properties under different pressure and temperature conditions. This premelting state represents something entirely different—not just another form of ice, but a hybrid phase that exists in the borderland between solid and liquid.
Future research will explore whether similar premelting states exist in other confined systems, from the narrow pores of geological formations to the tight spaces within biological membranes. The findings suggest that water’s behavior in confined spaces may be far more diverse and significant than previously imagined.
As Tadokoro notes, even substances as familiar as water continue to surprise us when examined under extreme conditions. The premelting state serves as a reminder that nature’s most fundamental processes still hold secrets waiting to be unlocked in the world’s smallest spaces.
Journal of the American Chemical Society: 10.1021/jacs.5c04573
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.