Every time you brace your core to stand up from a chair, tighten your abdomen before taking a step, or even tense up in anticipation of movement, something happens inside your skull that nobody had properly documented until now. Your brain shifts. Not dramatically, not in any way you could feel. We’re talking about a few microns, roughly the width of a red blood cell. But the movement is real, it’s measurable, and a new study published in Nature Neuroscience suggests it may be one of the reasons that walking around is good for your mind.
The study, from an interdisciplinary team at Penn State, overturns a long-held assumption: that the brain sits largely insulated from the mechanical forces of the body, cradled inside the skull, its movements governed by the familiar rhythms of heartbeat and breathing. In awake, moving animals, it turns out the dominant driver of brain motion isn’t the heart at all. It’s the stomach muscles.
The Pump Nobody Knew About
Patrick Drew, a professor of engineering science and neurosurgery at Penn State, has spent years trying to understand how the brain cleans itself. The organ lacks a conventional lymphatic system. Instead it depends on cerebrospinal fluid (CSF) washing through and around it, flushing out the metabolic waste that accumulates over a day of thinking. Sleep seems particularly important for this process, which is one reason scientists have linked poor sleep to higher risks of Alzheimer’s and other neurodegenerative diseases. But the mechanics of how CSF actually moves in an awake, behaving brain have been poorly understood. Nobody knew what, exactly, was driving the fluid around.
Drew’s team set out to watch. They fixed mice onto spherical treadmills, leaving the animals free to walk and run at will while two-photon microscopes tracked the precise position of the brain relative to the skull, frame by frame, at speeds faster than the cardiac cycle. Almost at once, a pattern emerged that surprised them. Brain motion didn’t track with breathing. It didn’t track with heartbeat. It tracked, tightly and reproducibly, with locomotion, and it started slightly before the mice began to move. Which meant whatever was causing the brain to shift, it wasn’t the locomotion itself.
The signal they were looking for, it turned out, was coming from the abdomen. The researchers implanted tiny electromyography electrodes into the abdominal muscles of the same mice, and found that the brain’s movement lagged the muscle contraction, consistently, by a fraction of a second. The abdominal muscles fired first. Then the brain moved. Not the legs, not the spine, not the neck. The gut.
A Hydraulic System Hidden in Plain Sight
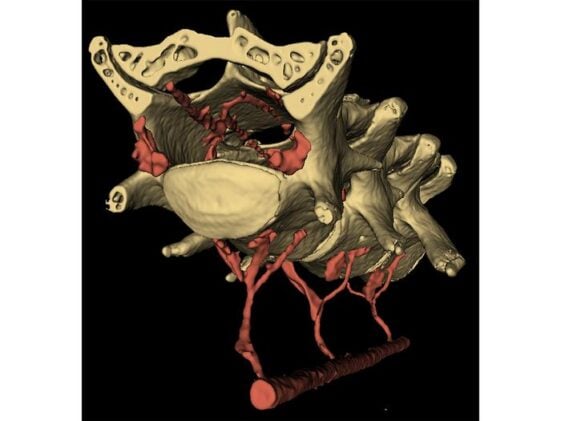
“In this study, we found that when the abdominal muscles contract, they push blood from the abdomen into the spinal cord, just like in a hydraulic system, applying pressure to the brain and making it move,” Drew said. The plumbing that makes this possible is a structure called the vertebral venous plexus, a network of valveless veins running between the abdominal cavity and the spinal canal. The VVP (as anatomists call it) is known in humans, where it’s thought to help buffer intracranial pressure during postural changes. Whether mice even had one was unclear until the team filled the vascular systems of two mice with a radiopaque compound and examined them with micro-CT imaging. The scans showed the plexus clearly, with small holes in the lumbar vertebrae through which veins thread between the abdominal cavity and the interior of the spinal column. When the abdominal muscles squeeze, they compress blood into this network. The blood moves into the spinal canal, narrows the dural sac surrounding the spinal cord, and drives cerebrospinal fluid cranially, pushing the brain forward and outward within the skull.
To confirm this mechanism wasn’t just an artifact of voluntary movement, the team wrapped lightly anaesthetised mice in a custom pneumatic belt placed around the abdomen and inflated it for two seconds. No limb movement, no postural change, just a gentle squeeze to the belly. The brain moved rostrally, just as it did during locomotion. “Importantly, the brain began moving back to its baseline position immediately upon relief of the abdominal pressure,” Drew said.
The Dirty Sponge Problem
Establishing that the abdomen moves the brain is interesting. Understanding what that movement does to fluid inside the brain is where the implications start to get genuinely consequential. Francesco Costanzo, a professor of engineering and mathematics also at Penn State, led the computational side of the project. Modelling fluid dynamics inside the brain is notoriously awkward (the organ is full of membranes and fluid compartments, all moving at once in ways that interact), so the team made a simplifying assumption. “The brain has a structure similar to a sponge, in the sense that you have a soft skeleton and fluid can move through it,” Costanzo said. With that framing in place, the computational question became almost domestic. “Keeping with the idea of the brain as a sponge, we also thought of it as a dirty sponge,” he added. “How do you clean a dirty sponge? You run it under a tap and squeeze it out.”
Their simulations, calibrated against the measured magnitudes of intracranial pressure and brain displacement from the mouse experiments, showed fluid flowing outward from the brain parenchyma into the surrounding subarachnoid space when the VVP squeeze was applied. The flows were large, several times the normal rate of CSF production, and they ran in the opposite direction from the glymphatic flows observed during sleep, when fluid enters the brain along the spaces surrounding penetrating arteries. That contrast fits neatly with a persistent puzzle in this field: why do tracer molecules infused into the CSF of sleeping animals enter the brain readily, while the same tracers in awake animals don’t penetrate the cortex at all? The simulations suggest an answer: during waking movement, the brain is being flushed outward. During sleep, it’s drawing fluid inward. Two complementary cycles, one running by night, one by day.
“Our research explains how just moving around might serve as an important physiological mechanism promoting brain health,” Drew said. The abdominal contractions involved are small, perhaps the sort you barely notice, a tightening of the core before any movement, the kind happening right now as you shift in your seat. “This kind of motion is so small. It’s what’s generated when you walk or just contract your abdominal muscles, which you do when you engage in any physical behavior. It could make such a difference for your brain health.”
There are obvious caveats. The study was done in mice. The mice were head-fixed, which removes the forces generated by head movement from the picture. The computational brain model is a simplified geometry, cylindrically symmetric, nothing like the actual wrinkled organ. The team doesn’t yet know how much of this transfers to humans, though the VVP is well documented in humans and abdominal pressure changes during exercise are, if anything, larger in our species. What they do flag, intriguingly, is that conditions that raise baseline intra-abdominal pressure, obesity, for instance, might chronically disrupt the normal hydraulic relationship between the gut and the brain, potentially contributing to the cognitive effects that have long been associated with excess body weight. Whether abdominal pressure also plays a role in the cognitive benefits reported after something as mundane as defecation (which sharply drops intra-abdominal pressure) is something the paper mentions, almost in passing, in the discussion section.
The deeper question the work opens up is how much of what we call “exercise benefits the brain” is really a story about pressure gradients in the abdomen and a network of veins that anatomists have known about for decades but nobody thought to connect to cognition. If the brain cleans itself partly through the mechanical consequences of moving your body, the implications stretch from basic neuroscience into clinical practice, exercise guidelines, and the design of therapies for people who can’t move. For now, though, the finding is simply this: your gut and your brain are in closer conversation than anyone suspected, and every step you take is part of it.
Garborg et al., Nature Neuroscience (2026). https://doi.org/10.1038/s41593-026-02279-z
Frequently Asked Questions
How does the gut physically move the brain?
When abdominal muscles contract, they raise pressure inside the abdominal cavity, compressing blood into a network of valveless veins called the vertebral venous plexus, which threads between the abdomen and the spinal canal through small holes in the lumbar vertebrae. This increased blood volume inside the spinal column narrows the dural sac surrounding the spinal cord, driving cerebrospinal fluid upward and pushing the brain forward by a few microns within the skull. The whole process works like a hydraulic pump, with the abdominal muscles as the pump and the vein network as the hydraulic line.
Is this movement large enough to actually matter?
A few microns sounds trivially small, but the fluid flows it generates are estimated to be several times larger than the normal rate at which the brain produces cerebrospinal fluid. That makes the movement potentially significant as a driver of fluid circulation through and around the brain, which is the mechanism thought to remove the metabolic waste products that accumulate during waking activity. Even very small physical displacements can produce outsized fluid-dynamic effects in a confined, pressurised space.
Does this explain why sleep is important for brain health?
Partly. The new simulations suggest that during waking movement, brain motion drives fluid outward from the brain parenchyma into the surrounding cerebrospinal fluid space. During sleep, a different process runs in the opposite direction: fluid enters the brain along spaces surrounding penetrating arteries, clearing waste more deeply into the tissue. The two cycles appear complementary, a daytime flushing and a nighttime deep-clean, which might help explain why both regular movement and good sleep seem independently important for long-term brain health.
Could obesity or a sedentary lifestyle disrupt this mechanism?
The researchers raise this as a possibility worth investigating. Obesity chronically elevates intra-abdominal pressure, which could alter the baseline state of the vertebral venous plexus and disrupt the normal pressure relationship between gut and brain. If the hydraulic cycle that moves cerebrospinal fluid is impaired, waste clearance could be less efficient, potentially contributing to the cognitive effects that have been observed in people with obesity. This remains speculative for now and hasn’t been tested directly.
Does this research apply to humans?
The study was conducted in mice, but the vertebral venous plexus is a well-established structure in humans, and abdominal pressure changes during exercise and movement are comparable in magnitude. The researchers are cautious about direct extrapolation, but the basic anatomy and hydraulics suggest the mechanism could operate similarly in people. Future work with human imaging during movement would be needed to confirm the connection, and to establish whether the fluid flows seen in simulations translate at the scale of the much larger human brain.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.