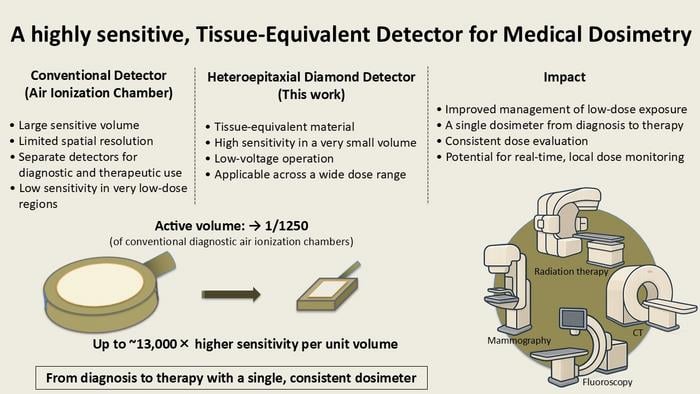
It sits on a fingertip. Four millimetres by four millimetres, half a millimetre thick, sealed inside a sliver of light-blocking polyethylene because even ordinary room light would throw off the readings. The device looks, for all practical purposes, like a fragment of nothing much. But this particular fragment of lab-grown diamond is roughly 1,250 times smaller than the radiation detectors routinely used in hospitals, and it can measure doses with a sensitivity per unit volume that exceeds those detectors by a factor of thousands. A team of physicists and engineers from Tokyo Metropolitan University, Tohoku University, and the materials company Orbray have just published the first systematic evaluation, and what they found is prompting some fairly serious rethinking of how medical facilities measure the radiation they deliver.
The problem the detector is meant to solve is one of those quiet clinical headaches that rarely makes news. Radiation dosimetry, the business of measuring exactly how much ionising radiation a patient or technician receives, underpins nearly everything in modern radiology and oncology.
The workhorse of the field is the air ionisation chamber, a device that works by detecting the tiny electrical current produced when X-rays knock electrons loose from gas molecules. It is reliable, it is calibrated, and it has been the gold standard for decades. The catch is that air isn’t especially good at absorbing X-rays, which means you need a fair volume of it to produce a measurable signal. Diagnostic X-rays, the kind used in ordinary chest and bone imaging, deliver doses at the low end of the clinical spectrum, and conventional air chambers struggle there. They’re bulky enough that mapping how dose varies across a region of tissue is impractical, and at very low exposures the signal barely clears the noise floor. Radiation therapy, meanwhile, operates at the other extreme, with doses many orders of magnitude higher. The result is that clinics often use entirely different instruments for the two settings. Which makes comparing measurements between them genuinely difficult, in a scientifically inconvenient sort of way.
Diamond, it turns out, has almost the ideal properties to fix this. Carbon has an atomic number close enough to the elements that dominate biological tissue, chiefly hydrogen, carbon, nitrogen, and oxygen, that a diamond detector responds to radiation in a way that closely mimics human flesh. This tissue equivalence is not a small thing.
The team grew their detector using a technique called heteroepitaxy, laying down diamond crystal atom by atom on a sapphire substrate using microwave plasma chemical vapour deposition. Earlier CVD diamond detectors were made from expensive single-crystal material, which limited how big and how cheap they could be. Heteroepitaxial growth sidesteps that constraint, allowing high-quality diamond to be produced over much larger areas; substrates up to 50 millimetres across have already been demonstrated. Once grown, the diamond was cut into a 4 by 4 millimetre chip, coated with titanium and gold electrodes on both faces, and connected to an electrometer. Before measurements began, the device had to be primed: irradiated with ten grays of therapeutic X-rays to fill the charge traps in the crystal lattice and bring it to a stable operating state. After that single conditioning step, the detector held steady.
Smaller Than a Fingernail, More Sensitive Than a Shoebox
What the team then demonstrated across a series of experiments was, by dosimetry standards, rather striking. At a bias voltage of just minus fifty volts, the diamond chamber reached a sensitivity comparable to a standard air ionisation chamber, despite being 1,250 times smaller by volume. Crank the voltage up to minus one hundred volts and the sensitivity doubles beyond the conventional reference. The energy dependence, how much the detector’s response shifts as X-ray photon energies change, stayed within ten per cent across the full diagnostic range. For a detector running without the energy-compensation filters that other small-volume instruments require, that’s genuinely flat. The team also showed excellent dose linearity: over the range of exposures typical in diagnostic radiology, the relationship between delivered dose and collected charge deviated from a straight line by less than three tenths of a per cent.
Monte Carlo simulations using the PHITS particle transport code confirmed that the raw physics supports the measurements, though the experimental sensitivity at higher voltages actually exceeded theoretical predictions. That gap probably reflects enhanced charge collection from interfacial electric fields near the electrode-diamond boundary and carrier dynamics involving trap states in the crystal bulk, though the exact mechanism isn’t yet fully pinned down. A productive mystery: the detector works better than theory currently explains.
The Consistency Problem No One Talks About
The institutional implication that the Tokyo team keeps returning to is consistency. Right now, “using the same device for dosimetry during both diagnosis and therapies could enable improved consistency,” as the university press release puts it. This matters more than it might seem. Clinical dose comparisons between treatment planning and delivery rely on a chain of measurements taken with instruments calibrated against different reference standards. Every link in that chain introduces error. A single detector platform that spans diagnostic and therapeutic energy ranges would shorten the chain considerably, and in radiological research, where scientists are trying to understand the biological effects of very low chronic exposures, the ability to compare doses across settings without translation factors could open up questions that have remained intractable.
The compactness of the device creates other possibilities too, some of them quite different in character. “It is compact enough to produce an array, like the sensor array on a camera, which could map the change in dose over some area,” the press release notes. In radiation therapy, dose mapping is critical: you want to know that the radiation field is hitting exactly the right shape, not just delivering the right total. Current detector arrays are bulky enough that spatial resolution suffers. A diamond array with 1,250 times better volumetric efficiency per element would let you pack detectors much more densely.
Some caveats are worth being clear about. This is a prototype, a single 4 by 4 millimetre chip tested under controlled laboratory conditions in Japan, not a product. The heteroepitaxial growth process is scalable in principle, but scaling from a research fabrication run to consistent clinical manufacturing is its own engineering challenge. The priming step, requiring ten grays of therapeutic irradiation before the device settles to a stable state, adds a logistical wrinkle that would need to be solved before routine deployment. And the delayed charge component seen at low doses and low voltages, a slow trickle of signal from carriers being released from crystal traps after the X-ray exposure ends, will need to be characterised more thoroughly for applications where precise temporal resolution matters.
None of that diminishes what the paper actually shows. The researchers have demonstrated something that nobody had managed before: a diamond solid-state ionisation chamber operating stably in the diagnostic X-ray energy range, at voltages low enough to be practical, with performance that rivals or surpasses conventional air chambers in a package that fits comfortably between your thumb and forefinger. The scalability of heteroepitaxial growth means the same device concept could eventually yield detectors with sensitivity more than a hundred times beyond anything available today.
In a field where the measurement tool is often what limits the science, that’s worth sitting with for a moment.
DOI / Source: 10.1002/mp.70363
Frequently Asked Questions
Why does it matter that diamond is similar to human tissue?
When a radiation detector responds to X-rays the same way biological tissue does, it doesn’t need complex correction factors to translate what it measures into a dose the body actually received. Most conventional detector materials absorb radiation differently from flesh, which means results have to be mathematically adjusted before they’re clinically meaningful. A near-tissue-equivalent detector like diamond gives you a more direct readout, and that simplicity becomes important when you’re trying to compare measurements across different clinical contexts.
Could this eventually replace air ionisation chambers in hospitals?
That’s the longer-term ambition, though there’s plenty of engineering still to do. The current device is a laboratory prototype that requires an initial conditioning dose of radiation before it settles to a stable state, and manufacturing consistency at clinical scale hasn’t been demonstrated yet. What the Tokyo team has shown is that the physics works, the sensitivity is there, and the fabrication method is in principle scalable. Whether that translates into a product depends on solving problems that are more about manufacturing than fundamental science.
What’s stopping researchers from measuring low-dose radiation effects right now?
The sensitivity limits of conventional detectors are a genuine bottleneck. Air ionisation chambers need a significant volume to accumulate enough signal at very low exposures, which limits how precisely you can localise a measurement. Studies of chronic low-dose radiation, of the kind relevant to occupational exposure or background environmental radiation, have historically struggled with instruments that aren’t sensitive enough to resolve the doses involved with confidence. A detector with thousands of times better volumetric sensitivity opens up experimental questions that have previously been out of reach.
Is a diamond radiation detector array something that already exists?
Not at the performance level this research suggests is achievable. Detector arrays are already used in radiation therapy for dose mapping, but the individual elements are relatively large, which limits spatial resolution. Because the diamond detector in this study achieves comparable sensitivity to a conventional chamber in a volume 1,250 times smaller, the elements of a diamond array could in principle be packed far more densely, producing much finer-grained dose maps. That’s still a research ambition rather than a product, but the geometry is plausible.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.