The obvious target, if you want to help someone stop smoking, seems like it would be the craving itself. Kill the desire, kill the habit. It’s the kind of logic that has driven decades of pharmacology aimed at the brain’s reward circuitry, with results that have been, at best, modest. Fewer than one in ten smokers achieves long-term abstinence even with the best available medications. Researchers at the Medical University of South Carolina took a different view. What if, instead of attacking craving directly, you strengthened the brain’s ability to resist it?
The answer, from a small but carefully designed clinical trial just published in the Journal of Psychiatric Research, suggests the distinction matters quite a lot.
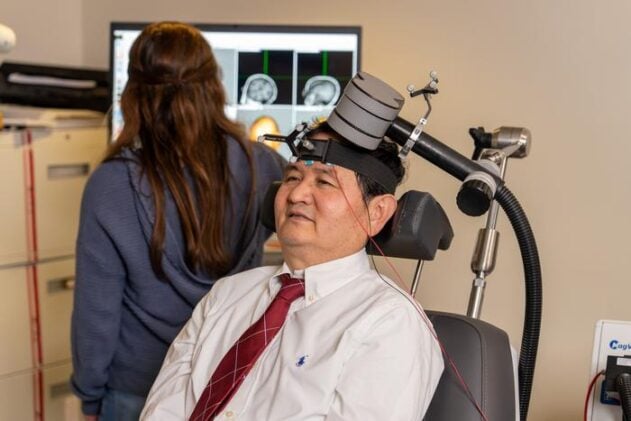
The treatment in question is repetitive transcranial magnetic stimulation, or rTMS, a technique that uses brief magnetic pulses to nudge cortical activity up or down depending on the frequency applied. It’s been approved for depression and OCD, and researchers have been probing its potential in addiction for some years now. The MUSC team, led by psychiatrist Xingbao Li, weren’t simply testing whether rTMS could help people smoke less. They were asking a more pointed mechanistic question: which neural circuit should you target, and why?
Two Strategies, One Decision
Addiction, in the neurobiological framing Li works within, is a kind of imbalance. “In addiction, brain systems can get out of balance,” he explained. “One system, tied to reward and craving, becomes overactive. Another, responsible for control and decision-making, becomes weaker.” That second system lives, broadly, in the dorsolateral prefrontal cortex, or DLPFC, a region at the front and side of the brain that is deeply involved in self-regulation. The reward system’s key node, for the purposes of this study, was the medial orbitofrontal cortex, a region involved in processing the value and salience of pleasurable things, including cigarettes.
Forty-six treatment-seeking smokers were randomised to one of three conditions. One group received high-frequency rTMS to the DLPFC, intended to excite that region and boost executive control. A second group received low-frequency rTMS to the orbitofrontal cortex, intended to dampen reward signalling. A third got sham treatment, a convincing imitation that produces the same sensations without the magnetic field reaching brain tissue.
Crucially, this wasn’t a one-size-fits-all protocol. Each participant received a structural MRI scan beforehand, with the resulting data used to compute how much electromagnetic field would actually reach their individual cortical surface. “It’s a kind of precision medicine,” Li said. “We’re tailoring the treatment to each person’s brain.” Over three weeks, participants came in five days a week for sessions that combined the stimulation itself with exposure to smoking-related cues, on the theory (supported by earlier work) that pairing rTMS with cue provocation amplifies the therapeutic effect.
The Prefrontal Cortex Wins
The results were not ambiguous. Participants in the DLPFC group reduced their cigarette consumption by an average of roughly 11 cigarettes a day, compared with around five in the orbitofrontal group and about six in the sham condition. Carbon monoxide levels in breath, a direct biological measure of how much someone is actually smoking, tracked the same pattern. Those effects, moreover, persisted through the one-month follow-up assessment, and appeared to strengthen slightly over time rather than fading.
The brain imaging data offered a window into why. After three weeks of DLPFC stimulation, participants showed increased activity in the prefrontal cortex during tasks requiring them to resist the urge to smoke, and reduced activity in the orbitofrontal region during cue-craving scans. The degree of change in each participant’s brain predicted, with some precision, how much their smoking had fallen. The orbitofrontal stimulation group, by contrast, showed no significant neuroimaging changes and no meaningful clinical benefit. “It’s a top-down effect,” Li said. “You enhance the control system, and it naturally regulates the reward system.”
This is not entirely surprising if you know the anatomy. The DLPFC has rich projections down into limbic and reward structures; turning it up seems to pull reward circuitry back under voluntary control rather than simply suppressing it. Trying to quiet the orbitofrontal cortex directly may, the authors speculate, blunt both the maladaptive craving-related activity and the adaptive processes that region supports, cancelling out any potential benefit.
What Comes Next
There are real limits to sit with here. Thirty-five people completed the trial, which is not many. The study was designed around the sensitive surrogate of cigarettes per day rather than full abstinence, meaning the headline figure of an 11-cigarette-a-day reduction, impressive as it looks, doesn’t yet translate cleanly to quit rates. The orbitofrontal and DLPFC conditions also differed in pulse frequency and total pulse number, which makes it harder to say with confidence that it was purely the target, rather than dose differences, driving the result.
Larger trials are underway. And the team now knows, at least directionally, where to point the coil. “We wanted to compare the approaches and pick a winner,” Li said. “Now we know which direction to take in future studies.”
The practical implications are sharpest for people who have already exhausted conventional options. Some smokers can’t tolerate varenicline. Others relapse repeatedly through counselling and nicotine replacement. Among people diagnosed with cancer, the numbers who keep smoking despite a diagnosis are striking, and the consequences severe. rTMS, if the larger trials bear out these initial findings, could offer something current pharmacology cannot: a direct intervention on the circuitry of self-control itself, rather than an approximation of it. The brain’s braking system, it turns out, may be the most useful lever we have. We just hadn’t been pulling it hard enough.
Source: Li et al., Journal of Psychiatric Research, 2026. DOI: 10.1016/j.jpsychires.2026.02.053
Frequently Asked Questions
Why didn’t targeting the craving part of the brain work?
The orbitofrontal cortex, which processes reward and craving, appears to serve more than one function, handling both the compulsive pull toward cigarettes and adaptive decision-making processes that support quitting. Suppressing it with low-frequency stimulation may therefore blunt helpful and harmful responses simultaneously, leaving no net benefit. Boosting the brain’s control system, by contrast, seems to bring reward circuitry into line without directly interfering with it.
How is this different from nicotine patches or varenicline?
Existing medications work primarily by mimicking or blocking nicotine at the receptor level, or by altering the dopamine signals that make smoking feel rewarding. rTMS doesn’t touch neurochemistry directly; it reshapes the functional activity of whole cortical circuits, specifically strengthening the prefrontal regions responsible for resisting impulses. Whether that makes it complementary or superior to pharmacotherapy is still an open question, but it represents a genuinely different mechanism of action.
Could this approach work for other addictions, not just smoking?
Possibly. The same prefrontal hypofunction and reward hyperactivation that characterises tobacco use disorder appears across alcohol, cocaine, and opioid addiction. DLPFC stimulation has been explored in several of these contexts, with mixed but generally encouraging results. The MUSC work adds to the case that strengthening executive control may be a transdiagnostic strategy, though each substance disorder has its own neural fingerprint and the evidence base varies considerably.
Is rTMS painful or risky?
In this trial, no serious adverse events occurred. The most commonly reported side effect was pain at the stimulation site, which subsided shortly after each session ended. rTMS is already approved and in clinical use for depression and obsessive-compulsive disorder, so its safety profile is reasonably well understood, though long-term effects in addiction treatment contexts remain less studied.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.