In a dimly lit lab in St. Louis, a mouse lies still. Its heart has slowed, its body temperature has dropped, and its cells have shifted into a low-energy state that echoes a wintering bear.
But this isn’t nature at work, it’s a scientist’s hand. Using a tiny, wearable ultrasound device, researchers have triggered a torpor-like state in the mouse’s brain. It’s artificial, precise, and, most crucially, reversible.
This is synthetic torpor, a long-imagined physiological pause button that could one day help doctors buy precious time — after a stroke, during an organ transplant, or even en route to Mars. A team led by Hong Chen, professor of biomedical engineering and neurosurgery at Washington University in St. Louis, is pioneering this work. Their method, detailed in Nature Metabolism, uses focused ultrasound to modulate the brain’s hypothalamus, coaxing the body into a deep, energy-saving mode without drugs, surgery, or genetic modification.
Learning from Nature’s Survival Artists
Torpor is common in the animal kingdom. Hummingbirds use it nightly to survive cold desert air. Bears rely on it to hibernate through harsh winters. But humans? We’ve never done it. That hasn’t stopped scientists from trying.
“Synthetic torpor is no longer just a theoretical concept,” said Chen. “It is an emerging field with the potential to redefine medicine.”
Chen’s group is the first to demonstrate a noninvasive, reversible method of inducing this state in both mice and rats. While mice naturally enter torpor, rats do not — yet the technique worked in both. That opens the door to future human applications, where torpor could protect the brain and organs by slowing the body’s metabolic machinery instead of trying to rev it back up.
Dialing Down the Body’s Demands
Traditional medicine often focuses on restoring energy supply — think of the rush to open blocked arteries after a heart attack. Synthetic torpor flips that logic: reduce the body’s demand for energy, and you give it time to recover. During induced torpor, body temperature in mice dropped about 3°C. Heart rates slowed nearly 50%, and metabolism shifted entirely to fat burning, mirroring natural torpor’s signature energy mode.
“The capability of synthetic torpor to regulate whole-body metabolism promises to transform medicine,” Chen and colleagues wrote in a July 31 Perspectives piece in Nature Metabolism.
The Real-World Possibilities
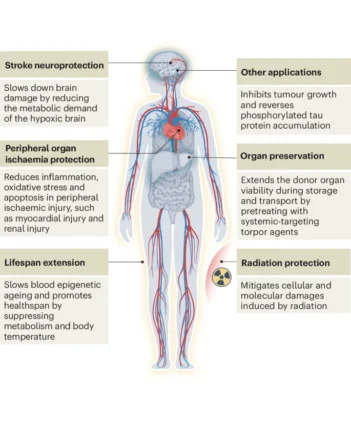
The implications stretch far beyond the lab. In preclinical models, synthetic torpor has:
- Reduced brain damage in mice by 71% after simulated strokes
- Preserved organ function during low blood flow or transplant-like conditions
- Protected against radiation-induced tissue injury
- Slowed biological aging markers in long-term experiments
There’s even evidence that inducing torpor can influence tau protein accumulation, a key driver of Alzheimer’s disease, and may suppress tumor growth in some cancer models.
Challenges Between Mice and Mars
For all its promise, synthetic torpor isn’t ready for the clinic — or the cosmos — just yet. Human bodies are larger, slower to cool, and far more complex than rodents. Past attempts using compounds like hydrogen sulfide ran into dangerous side effects. But ultrasound may offer a safer route forward. It’s already FDA-approved for other brain therapies, and the WashU team’s method is completely noninvasive.
Wenbo Wu, a doctoral student in Chen’s lab and lead author on the recent paper, emphasized the hurdles ahead: differences in metabolism between species, the need for reversible control, and the ethical considerations of using torpor in critical care. “Collaboration among scientists, clinicians and ethicists will be critical,” Wu said.
What Comes Next?
The next frontier involves testing these methods in larger mammals and fine-tuning the balance between central brain control and peripheral metabolic systems. Chen’s team envisions a dual approach: using brain stimulation to induce torpor while also delivering drugs or modulating nerves outside the brain to sustain it safely.
It’s not lost on the team that science fiction has long imagined this scenario: astronauts in cryosleep, humans preserved for healing or space travel. The difference now is that it no longer seems so far-fetched.
“Realizing this potential will require not only bridging the gaps between neuroscience, engineering and medicine,” Chen wrote, “but also building an inclusive research ecosystem, securing sustained funding and engaging the public in thoughtful dialogue.”
A New Kind of Emergency Medicine
Picture this: a paramedic places a device on a trauma victim’s forehead, presses a button, and buys two hours of metabolic stillness before surgery. Or an astronaut headed to Jupiter quietly slips into torpor, using only a fraction of normal oxygen and food. These are not tomorrow’s dreams — they are today’s goals.
And it all starts with a mouse, lying quietly in a lab, waiting to wake up.
Journal: Nature Metabolism
DOI: 10.1038/s42255-025-01345-3
Authors: Wenbo Wu, Genshiro A. Sunagawa, Hong Chen
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.