Key Takeaways
- Oligodendrocytes retract instead of dying under stress, leading to potential implications for myelin regeneration.
- A study found that dasatinib and quercetin significantly demyelinated mouse brains without killing cells, surprising researchers.
- The effect was worse in younger mice, suggesting they may depend more on energy pathways affected by D+Q.
- D+Q-treated cells resembled immature oligodendrocytes found in multiple sclerosis, indicating possible reversibility of demyelination.
- This research raises concerns for off-label use of D+Q and highlights a potential pathway for studying recovery in neurodegenerative diseases.
The oligodendrocyte is a strange-looking cell. It sends out dozens of thin processes in every direction, wrapping each one around a nearby nerve axon like a spiral of electrical tape. The resulting sheath is myelin, and without it, nerve signals stall, slow, and eventually fail. An oligodendrocyte at full function is architecturally complex, almost baroque, a single cell tending to the insulation of perhaps fifty axons at once. An oligodendrocyte under stress does something unexpected. It doesn’t die. It simply retracts.
That detail is now at the center of an unexpected finding from the University of Connecticut, published Monday in the Proceedings of the National Academy of Sciences. A two-drug combination called dasatinib and quercetin, which has become one of the most widely tested interventions in anti-aging research, causes significant demyelination in the brains of mice. Both young and old. Without killing a single cell.
Dasatinib and quercetin, usually shortened to D+Q, belong to a class of drugs known as senolytics. The concept is straightforward enough: accumulating senescent cells drive inflammation and accelerate aging; clear them out pharmacologically and the body does better. Evidence for this has been building for years, and D+Q is now in clinical trials for conditions ranging from type 2 diabetes to Alzheimer’s disease. Some people in the anti-aging community use it off-label. The medical establishment is cautious about that, though until recently the concern had focused on peripheral effects, not the brain. Very few studies had looked at what senolytics actually do to the central nervous system.
Evan Lombardo and Robert Pijewski, then working in immunologist Stephen Crocker’s lab at UConn School of Medicine, set out to answer a more hopeful question entirely. They wanted to know whether D+Q could rejuvenate the brains of mice with multiple sclerosis, potentially healing the damage that accumulates over years of the disease. They tried it first in healthy animals, both young (around six to nine months) and aged (twenty-two months), expecting a baseline. What they got instead was the finding they’d been hoping to avoid.
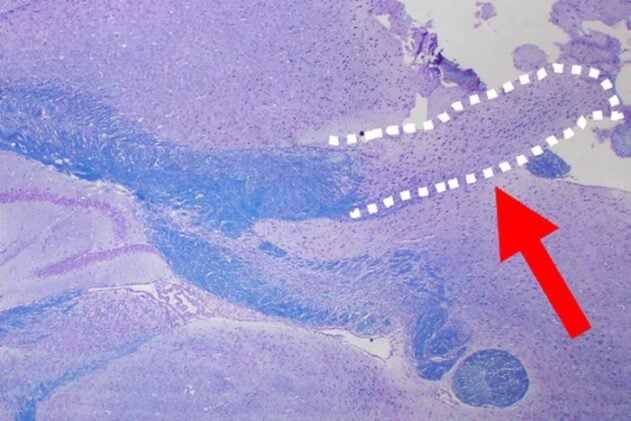
Healthy mice have myelin visible under the microscope as dark rings around lighter axons, a consistent pattern of tight insulation. The D+Q-treated animals had far less of it. The corpus callosum, the thick band of white matter connecting the two cerebral hemispheres, was visibly affected, a region whose damage is associated with the cognitive and motor symptoms popularly known as “chemo brain.” The effect was worse in younger animals than in older ones, which surprised the team considerably.
Crocker notes that the damage appeared in animals of every age tested, and was, if anything, more severe in the younger ones.
The researchers then took the investigation into cell culture, treating oligodendrocyte progenitor cells with D+Q in a lab dish and filming what happened. Time-lapse video of the treated cells shows something almost melancholy: processes withdrawing one by one, complexity collapsing inward, the cell still alive but reduced to a simpler version of itself. Control cells, treated with vehicle only, maintained their architecture across the same period. When the team ran bulk RNA sequencing on the D+Q-treated oligodendrocytes, they found a cluster of differentially expressed genes pointing toward endoplasmic reticulum stress, the cellular equivalent of a production line jamming under load. The most plausible culprit is the unfolded protein response, a stress pathway the cell activates when protein production begins to overwhelm quality control. “We suspect the drugs are choking off energy the cells need,” Crocker says, “and the cells respond by reducing complexity, reverting to a younger state, but less functional.”
Here the story takes its more consequential turn. The reverted cells, sitting in a dish having been treated with anti-aging drugs, looked strikingly like a known population of oligodendrocytes found in the brains of people with multiple sclerosis. Not dead cells. Immature ones. Cells that have apparently retreated to an earlier developmental stage under stress, shedding the myelinating function they were supposed to maintain. If that parallel holds, it suggests something about MS that the field has struggled to articulate cleanly: that demyelination in the disease may not primarily be about oligodendrocyte death, but about dysfunction, regression, perhaps something that is in principle reversible.
The caveat here is that this is mouse data from healthy animals, not a human MS cohort, and the mechanism remains proposed rather than demonstrated. The unfolded protein response is suggestive, not proven as the causative link. Clinical trials of D+Q are ongoing, and none have yet reported neurological damage in participants, though the team notes that measuring myelin loss in humans requires careful attention and is not always a primary endpoint in trials focused on systemic aging markers.
Still, the implications for the tens of thousands of people who use D+Q off-label are serious enough to warrant caution, particularly given the counterintuitive direction of the effect. You might expect aged myelin to suffer most from a metabolic insult. Younger oligodendrocytes, it turns out, may depend more heavily on precisely the energy pathways these drugs disrupt.
What the lab is working on now is the recovery question, which is where the accidental model becomes genuinely useful. Because the oligodendrocytes didn’t die, because they merely regressed, there is at least a possibility that the right intervention could bring them back. The cellular machinery for myelination is presumably still present, dormant rather than gone. Understanding why D+Q sends oligodendrocytes into retreat might illuminate why the same thing happens in MS, and potentially how to reverse it in both contexts.
Whether the drug community will welcome that irony, that one of anti-aging medicine’s most promising interventions turns out to offer a new way of modeling one of neurology’s most treatment-resistant diseases, remains to be seen. But the oligodendrocytes, quietly withdrawing in their dishes, may have something useful to say about repair.
DOI / Source: https://doi.org/10.1073/pnas.2524897123
Frequently Asked Questions
The combination is currently being tested in multiple clinical trials for conditions including type 2 diabetes and Alzheimer’s, but this new research suggests that its effects on the brain have been substantially underexplored. The UConn study found significant myelin damage in both young and old mice at doses used in aging research, with the effect worse in younger animals. Until larger human trials measure neurological endpoints systematically, the medical community’s existing caution about off-label use looks considerably more justified than it did before.
The working hypothesis is that dasatinib and quercetin interfere with energy metabolism in oligodendrocytes, the cells that produce and maintain myelin, triggering a stress response that causes the cells to retract their processes and regress to a simpler state. Crucially, the cells don’t die; they effectively de-differentiate, shedding function without disappearing. The same stress pathway, the unfolded protein response, is associated with endoplasmic reticulum overload and has been implicated in several neurodegenerative conditions.
Possibly, though not in the way originally intended. The regressed oligodendrocytes produced by D+Q treatment look strikingly similar to dysfunctional cells found in MS lesions, giving researchers an unexpected laboratory model for studying why myelin fails to regenerate in the disease. Because the cells remain alive in their immature state, there’s a plausible pathway to studying recovery, and the UConn team is now investigating whether the cells can be coaxed back into full myelinating function.
Myelin acts as insulation around nerve axons, allowing electrical signals to travel quickly and efficiently. When it degrades, signals slow, stall, or misfire, producing the numbness, pain, coordination problems, and cognitive symptoms characteristic of MS. The corpus callosum, the region most visibly affected in the mouse study, connects the two hemispheres of the brain and is involved in integrating information across motor, sensory, and cognitive systems. Damage there can cause the cluster of symptoms sometimes called “chemo brain.”
Counterintuitively, no. The demyelination was actually worse in younger mice than in the aged ones, which the researchers found surprising. The current hypothesis is that younger oligodendrocytes may rely more heavily on the specific metabolic pathways disrupted by D+Q. If that proves true in further work, it would have significant implications for the use of senolytics in younger people pursuing preventive anti-aging interventions.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.