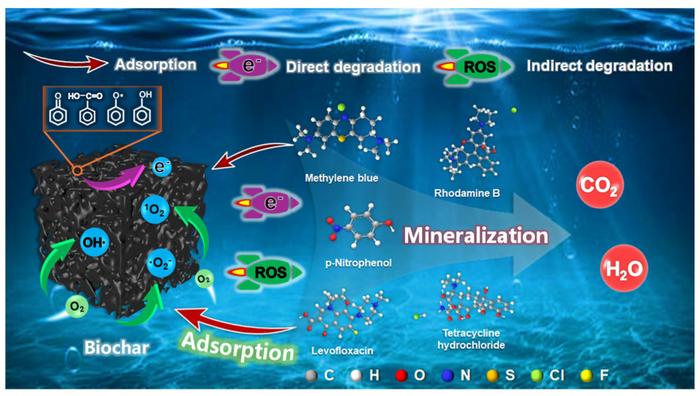
For decades, researchers believed biochar cleaned contaminated water like a simple sponge, passively absorbing pollutants from wastewater. But a groundbreaking study from Dalian University of Technology reveals this carbon-rich material has been quietly operating as something far more sophisticated: an electron-powered pollutant destroyer that actively breaks down contaminants without requiring additional chemicals.
Dr. Yuan Gao and his research team discovered that biochar doesn’t just trap harmful substances—it eliminates them through direct electron transfer, a natural ability that has been overlooked for years. Their findings, published in Carbon Research, demonstrate that this direct degradation process accounts for up to 40% of biochar’s total pollutant removal capacity.
The Hidden Power of Electron Transfer
The revelation came through advanced electrochemical testing and quantification methods that separated biochar’s various cleaning mechanisms. While previous research focused on biochar’s role as either an adsorbent or a catalyst requiring oxidants like hydrogen peroxide, this study identified a third pathway operating independently.
“Biochar has been underestimated. It’s not just a sponge—it’s a battery, a conductor, and a degrader all in one. We’re just beginning to tap into its true potential.”
The research team tested five different types of organic pollutants, including pharmaceuticals, dyes, and phenolic compounds. Using liquid chromatography-mass spectrometry analysis, they confirmed that biochar was actually transforming these contaminants into different molecular structures rather than simply holding them in place.
The secret lies in biochar’s structural features. Three key characteristics enable this electron-transfer superpower: carbon-oxygen (C-O) functional groups that serve as “handholds” for electron transfer, oxygen-hydrogen (O-H) groups that facilitate the process, and graphitic carbon structures that create “highways” for electrons to travel efficiently.
Game-Changing Implications for Water Treatment
This discovery fundamentally shifts how biochar could be deployed in water purification systems. Traditional advanced oxidation processes rely heavily on chemical additives to generate reactive oxygen species that break down pollutants. The new findings suggest biochar can accomplish similar results using only its inherent electron-donating capacity.
The practical implications are substantial. Water treatment facilities could potentially reduce their chemical consumption while maintaining effective pollutant removal. The process generates less sludge and operates without requiring additional oxidants, making it more environmentally sustainable and cost-effective.
Perhaps most impressively, the biochar maintained its direct degradation capability even after five reuse cycles, showing nearly 100% stability. This durability addresses one of the major challenges in water treatment: maintaining consistent performance over extended periods.
The research also revealed interesting competitive dynamics in natural water systems. Dissolved oxygen competes with organic pollutants for electrons from biochar, reducing direct degradation efficiency by 30-70% compared to oxygen-free conditions. This finding helps explain why previous studies may have underestimated biochar’s degradation potential.
“The direct degradation capacity of biochar was highly related to the electron-donating capacity.”
The team’s work involved creating modified biochar samples using ammonium polyphosphate at various ratios, producing materials with tailored characteristics. The most effective sample achieved direct degradation rates of 11 milligrams per gram of biochar—performance comparable to expensive iron-based catalysts but without the associated metal leaching risks or need for acidic conditions.
Beyond the laboratory, this research opens possibilities for more sophisticated biochar engineering. By understanding exactly which structural features drive electron transfer, researchers can now design biochar with enhanced direct degradation capabilities for specific pollutant types.
The study also identified degradation intermediates, confirming that pollutants like p-nitrophenol transform into compounds such as p-aminophenol and phenol during the electron transfer process. This detailed molecular understanding provides crucial information for predicting treatment outcomes and optimizing system design.
While challenges remain—particularly with high redox potential pollutants and complex real-world wastewater compositions—this research establishes a new foundation for biochar applications in environmental remediation. The findings suggest that biochar’s role in water treatment has been significantly undervalued, with direct electron transfer representing an untapped mechanism for pollutant removal.
As global industrial pollution continues threatening water resources, discoveries like this offer blueprints for more effective, sustainable treatment technologies. The research transforms biochar from a passive filtration material into an active environmental warrior, silently zapping contaminants one electron at a time.
Carbon Research: 10.1007/s44246-025-00219-3
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.