Korean scientists have mapped the cellular journey that transforms benign brain tumors into aggressive, treatment-resistant cancers, potentially opening new avenues for preventing deadly recurrences.
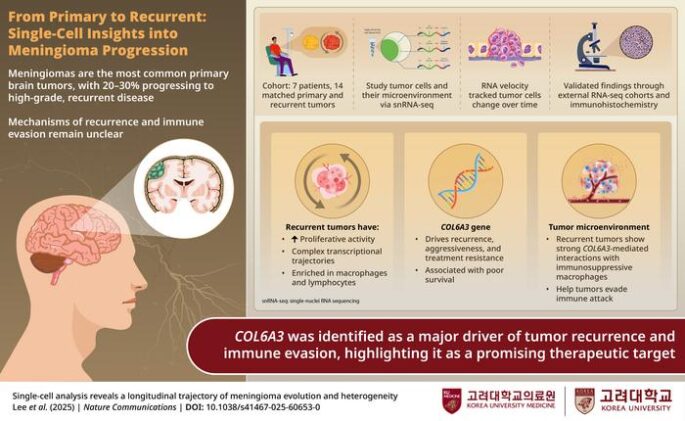
The research, published in Nature Communications, represents the first comprehensive analysis of how meningiomas evolve between their initial appearance and their often-fatal return. Using cutting-edge single-cell sequencing technology, researchers from Korea University tracked tumor cells and immune responses across 14 matched pairs of primary and recurrent tumors from seven patients.
Hidden Complexity Emerges at Recurrence
Meningiomas account for nearly one-third of all brain tumors, with most remaining manageable throughout a patient’s lifetime. However, 20 to 30 percent progress to high-grade forms that resist standard treatments and return with a vengeance. The cellular mechanisms driving this transformation have remained largely mysterious until now.
The Korean team discovered that recurrent tumors don’t simply grow faster than their predecessors. Instead, they undergo fundamental changes in their cellular programming, switching on proliferation pathways while simultaneously recruiting immunosuppressive cells that help shield them from the body’s natural defenses.
“Our study generated the first longitudinal single-cell atlas of matched primary and recurrent meningiomas. This resource enabled us to reconstruct tumor evolutionary trajectories and cellular hierarchies over time, revealing profound shifts in proliferative programs and tumor-immune interactions at recurrence.”
The researchers used RNA velocity analysis to track how individual tumor cells change their gene expression patterns over time. Primary tumors showed relatively stable, predictable growth patterns. Recurrent tumors, by contrast, exhibited chaotic, multidirectional changes that culminated in highly aggressive cellular states.
At the molecular level, one protein emerged as a central player in this transformation: COL6A3, part of the extracellular matrix that provides structural support around cells. In recurrent tumors, COL6A3 levels surged dramatically, and laboratory experiments confirmed its direct role in driving tumor cell proliferation.
Immune System Betrayal
Perhaps more troubling was the discovery of how recurrent tumors manipulate their surrounding environment. The researchers found that COL6A3 doesn’t just make tumor cells grow faster – it also recruits specific types of immune cells called C1Q+ macrophages that actually protect the tumor rather than attack it.
“We identified COL6A3 as a key driver of meningioma recurrence, associated with relapse risk and treatment resistance. Further analysis of tumor cell-macrophage interactions revealed that the COL6A3-CD44 signaling cascade mediates extracellular matrix remodeling and promotes an immunosuppressive tumor microenvironment at recurrence.”
This finding helps explain why recurrent meningiomas often prove so difficult to treat with conventional therapies. The tumors essentially hijack the immune system, turning would-be defenders into collaborators that help cancer cells survive and spread.
The research team validated their findings across multiple independent patient datasets, consistently finding elevated COL6A3 levels in recurrent tumors. When they artificially reduced COL6A3 levels in laboratory-grown meningioma cells, proliferation dropped significantly and cell division cycles slowed.
Current treatment approaches for meningiomas rely heavily on surgical removal followed by radiation therapy for high-grade cases. However, these methods fail to address the underlying cellular changes that drive recurrence, leaving patients vulnerable to tumor return months or years later.
The identification of COL6A3 as a key driver opens possibilities for more targeted interventions. Researchers suggest the protein could serve dual purposes: as a biomarker to identify high-risk patients earlier, and as a therapeutic target for drugs designed to block its tumor-promoting effects.
The work also reveals the importance of considering both tumor cells and their surrounding microenvironment when developing treatments. Traditional cancer therapies often focus exclusively on killing tumor cells, but this research suggests that disrupting the communication between cancer cells and immune cells might prove equally important.
Looking ahead, the research team anticipates their findings could impact patient care within the next five to ten years by enabling better prediction of which patients will respond to radiation therapy and which face higher recurrence risks. Such tools could help doctors tailor treatment approaches more precisely to individual patients.
The study analyzed tumors from patients ranging from grade I benign meningiomas to grade III anaplastic forms, with follow-up periods extending several years after initial treatment. All patients had undergone craniotomy for tumor removal, with an average age of 50 years at diagnosis.
While the research provides crucial insights into meningioma biology, translating these discoveries into clinical applications will require additional studies to develop and test COL6A3-targeted therapies. The complexity of the tumor microenvironment means that successful treatments will likely need to address multiple pathways simultaneously rather than targeting single molecules in isolation.
Nature Communications: 10.1038/s41467-025-60653-0
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.