Nature does it with ease, but chemists have struggled. A Swiss team has now shown that a single molecule can grab two photons in sequence and store two oxidations and two reductions without chemical crutches.
The study, published August 25 in Nature Chemistry, demonstrates a donor–photosensitizer–acceptor pentad that traps a double charge state with 37 percent efficiency and holds it for up to 120 microseconds. The system stores 3.0 electron volts of energy, an encouraging step toward artificial photosynthesis.
The Long Road From One Electron To Many
Photosynthesis in plants relies on moving several electrons to make fuels like sugars. Lab systems, in contrast, tend to move only one electron at a time. To bridge the gap, chemists often add sacrificial reagents that push reactions in one direction but are wasted in the process. The Basel team wanted a cleaner way.
The Pentad Design
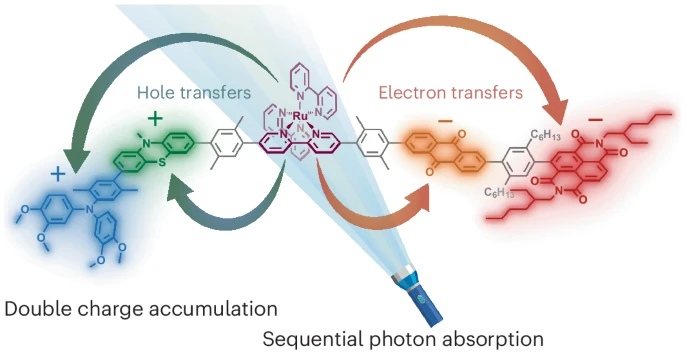
Their molecule has five linked parts: two electron donors, a ruthenium bipyridine complex that soaks up light, and two electron acceptors. One photon triggers the first charge split, sending an electron across the structure and creating a long-lived state. A second photon re-excites the ruthenium center. This time, fresh charges appear on the second donor and acceptor, leaving the molecule with four redox equivalents neatly parked on its branches.
“Our design principle leads to an unprecedented scenario in which the charge accumulating step proceeds with the same high quantum yield as the initial charge separating step,” the authors wrote in Nature Chemistry.
Testing The Idea
The researchers used laser pump–probe experiments to track the charged states in real time. First they saw the single electron–hole pair. Then, by keeping molecules in that state with a continuous laser and hitting them again with a pulse, they watched a second pair appear. The signals matched the expected fingerprints of the new charged units.
The result: a double charge state with a lifetime of at least 100 nanoseconds, probably longer. That makes it one of the most stable examples of its kind.
Why It Matters
Earlier attempts required blasting molecules with light far stronger than sunlight, often with two sensitizers absorbing at once. This design works under weaker light and uses sequential excitation, which is much closer to what real solar energy provides. It opens the way for molecules that could one day drive water splitting or carbon dioxide reduction directly.
Key Findings
- Team: Mathis Brändlin, Björn Pfund, Oliver S. Wenger, University of Basel
- Journal: Nature Chemistry, August 25, 2025
- System: Five-part molecule with two donors, one photosensitizer, two acceptors
- Quantum yield: 37 percent for double charge state
- Lifetime: 100 nanoseconds to 120 microseconds
- Energy stored: 3.0 electron volts
- Significance: Sequential light absorption at low irradiance, no sacrificial reagents
Takeaway
A molecular pentad can now accumulate two positive and two negative charges with ordinary light, a feat long out of reach. The advance marks an important step toward artificial systems that might someday turn sunlight into fuels.
Journal: Nature Chemistry, August 25, 2025.
DOI: 10.1038/s41557-025-01792-8
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.