Mice under stress give up faster. They stop swimming, stop struggling. It’s a crude proxy for despair, but for decades it’s been how researchers screen antidepressants. And for decades, one promising brain target kept failing that test in humans, even though it worked beautifully in rodents.
The target was neurokinin-1 receptor, or NK1R. By the early 2000s, after drugs like aprepitant flopped in clinical trials, most researchers concluded the biology was wrong. Korea University scientists now say the chemistry was the problem instead.
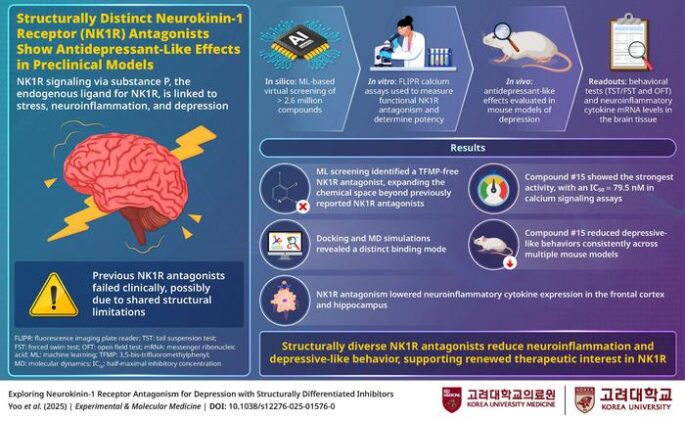
In a study published in Experimental & Molecular Medicine, the team reports that structurally novel NK1R inhibitors reduce depressive behavior and brain inflammation in mice. The key wasn’t abandoning the target. It was abandoning a chemical feature nearly every previous drug shared.
One Scaffold, Many Failures
Most earlier NK1R antagonists contained something called a 3,5-bis-trifluoromethylphenyl group. That motif helps drugs grip the receptor tightly, but it may also create problems elsewhere, affecting how the body metabolizes compounds or how they interact with other proteins.
The Korea University team stripped it out entirely. Using machine learning to screen millions of molecules, they identified candidates with fundamentally different structures. One, labeled compound #15, stood out.
It bound NK1R in a distinct way and worked across multiple mouse models. Animals exposed to social isolation, restraint stress, or inflammatory challenges showed less immobility in behavioral tests. They didn’t give up as fast. At the same time, markers of neuroinflammation dropped in brain regions tied to mood, including the frontal cortex and hippocampus.
That anti-inflammatory effect matters. Chronic inflammation is one reason some patients don’t respond to standard antidepressants like SSRIs.
“Our findings suggest that the structurally distinct antagonists identified in this study exhibit antidepressant-like effects, providing renewed evidence for further exploration of NK1R antagonism as a therapeutic strategy for MDD,” Hong-Rae Kim explains.
A Different Fit
Computer modeling revealed compound #15 forms a unique hydrogen bond inside the receptor pocket, one that older drugs couldn’t manage. That altered interaction may explain why it produces effects the earlier generation didn’t.
The study doesn’t claim NK1R antagonists are ready for patients. Results come entirely from animal models, and translation to humans has been the graveyard for many depression drugs. Safety and pharmacokinetics remain unknown. Whether these compounds would beat existing treatments is an open question.
But the work reframes a 20-year-old disappointment. When a drug class fails clinically, it’s easy to assume the biological target was a mirage. This suggests the molecules themselves might have been the limiting factor.
For depression research, that’s not a small distinction. Progress may sometimes come not from discovering new targets, but from learning how to interrogate old ones with better chemistry.
DOI: https://doi.org/10.1038/s12276-025-01576-0
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.