Scientists have created an optical biosensor that identifies cancer DNA in blood samples within 20 minutes, achieving near-perfect accuracy in distinguishing between healthy individuals and cancer patients.
The device, developed at the Korea Institute of Materials Science, detects methylated DNA — chemical changes that occur when cancer develops — at concentrations 1,000 times lower than current methods allow. In tests with 60 colorectal cancer patients, the sensor correctly identified cancer presence 99% of the time.
How the Technology Works
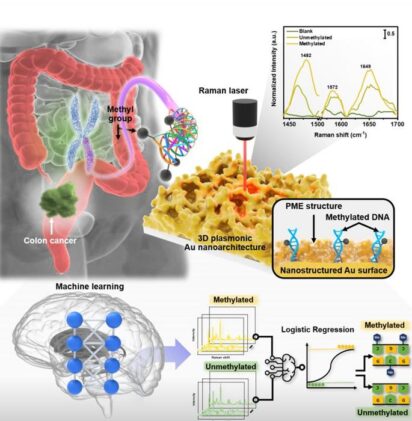
The biosensor uses plasmonic materials that amplify light signals from DNA molecules by more than 100 million times. When laser light hits these materials in the presence of cancer DNA, they create electromagnetic “hotspots” that enhance normally weak molecular signals.
The sensor can detect methylated DNA at just 25 femtograms per milliliter — equivalent to finding 1/25,000th of a sugar grain dissolved in a water drop.
Key Advantages Over Current Methods
- Requires only 100 microliters of blood (about two drops)
- Delivers results in 20 minutes versus hours or days
- No complex sample preparation needed
- Distinguishes between cancer stages I through IV
- Achieves 100% sensitivity and 98.3% specificity
The technology employs a process called plasmonic molecular entrapment (PME). Gold nanostructures trap DNA molecules while simultaneously growing additional gold layers around them, creating optimal conditions for signal amplification.
Machine Learning Enhances Accuracy
An artificial intelligence algorithm analyzes the light patterns to determine methylation levels. The logistic regression model examines specific wavelengths where methylated DNA produces distinct signals, particularly at 1,482 cm⁻¹ on the Raman spectrum.
The AI system quantifies global methylation levels — the overall percentage of methylated DNA across the genome. Cancer patients showed lower methylation (12.7%) compared to healthy individuals (15.9%), reflecting a phenomenon called global hypomethylation associated with tumor development.
Clinical Performance Details
The researchers tested serum samples from 40 colorectal cancer patients across all stages and 20 healthy controls. Beyond simple detection, the sensor differentiated cancer stages with remarkable precision:
- Stage I: 14.4% methylation (86.7% detection sensitivity)
- Stage II: 13.1% methylation (80% detection sensitivity)
- Stage III: 12.3% methylation (90% detection sensitivity)
- Stage IV: 11.2% methylation (93.3% detection sensitivity)
The decreasing methylation levels correlate with cancer progression, providing insights into disease severity. Five-fold cross-validation confirmed the model’s reliability, with accuracies ranging from 81.5% to 93.8% across different patient groups.
Technical Innovation: Interparticle Gaps
High-resolution imaging revealed a crucial detail: the sensor creates nanoscale gaps of 2.52 nanometers between gold particles when DNA is present. These gaps generate intense electromagnetic fields precisely where DNA molecules sit, maximizing signal enhancement.
The team verified this mechanism using electron energy loss spectroscopy, which showed nitrogen atoms from DNA uniformly distributed across the sensor surface. Real-time monitoring captured the signal amplification process occurring over 280 seconds as gold structures formed around trapped DNA.
Future Applications
While initially developed for colorectal cancer, the platform could potentially detect various cancers and diseases involving DNA methylation changes. The researchers envision applications in hospitals, screening centers, and even home diagnostic kits.
“This technology serves as a next-generation diagnostic platform capable not only of early cancer detection, but also of predicting prognosis and monitoring treatment response,” states Dr. Ho Sang Jung, Senior Researcher at KIMS and lead of the project.
The sensor’s ability to work with raw serum samples eliminates time-consuming DNA extraction steps required by conventional methods. Combined with its compact design and rapid analysis, this positions the technology for point-of-care testing in clinical settings.
The research appears in the May 2025 issue of Advanced Science.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.