Scientists have pinpointed a rare type of brain cell that goes haywire in mice showing schizophrenia-like behavior, and when they dialed down the activity of these cells, the animals’ symptoms improved. The discovery suggests a narrow window during brain development when doctors might one day intervene to stop cognitive problems before they start.
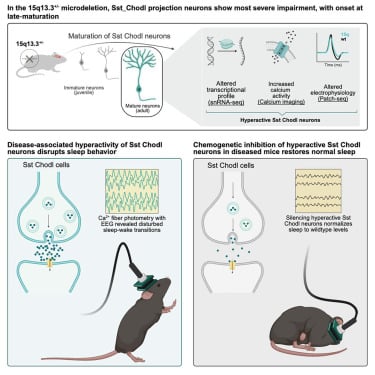
The cells in question are a subset of inhibitory neurons called Sst_Chodl cells. They make up only a tiny fraction of the brain’s cellular landscape, but they wield outsized influence over how neural circuits operate. In mice carrying a genetic deletion linked to schizophrenia in humans, these cells became abnormally active just as the animals transitioned from adolescence to adulthood, and their sleep patterns fell apart.
“Current treatments for cognitive symptoms in patients with diagnoses such as schizophrenia are inadequate. We need to understand more about what causes these cognitive symptoms that are derived from impairments during brain development,” says Konstantin Khodosevich, a professor at the University of Copenhagen’s Biotech Research and Innovation Center. “Our study may be the first step toward a new, targeted treatment that can prevent cognitive symptoms.”
A Chain That Snaps
Schizophrenia doesn’t announce itself at birth. The disorder originates in faulty brain wiring that begins during fetal development, yet symptoms like hallucinations, delusions, and cognitive fog typically emerge only in late adolescence or early adulthood. For years, researchers have puzzled over this delay. Why does the brain tolerate developmental errors for so long before collapsing into dysfunction?
Katarina Dragicevic, one of the study’s first authors, traced brain development from the prenatal stage all the way to adulthood in mice with a deletion of chromosome region 15q13.3. In humans, this same genetic glitch is associated with epilepsy, autism, and schizophrenia. She found that molecular and functional changes remained relatively modest through childhood. Then, as the animals hit adolescence, the system tipped.
“For a long time, the brain is able to compensate for developmental errors and maintain relatively normal function. But at some point, it’s like a chain snapping: the brain can no longer compensate, and that’s when symptoms emerge,” Dragicevic explains. “Until that point, however, prevention should be possible.”
That pre-snap window could be the sweet spot for intervention. If doctors could identify children at risk and stabilize their brain circuits before the chain breaks, they might head off years of debilitating symptoms.
Sleep as a Window into Brain Chaos
Sleep disturbances are common in people with psychiatric disorders, so the Copenhagen team used sleep as a behavioral readout. They monitored the mice around the clock, tracking when they dozed off, how deeply they slept, and how often they woke. The mutant mice slept poorly, waking frequently and spending less time in restorative slow-wave sleep.
Using single-nucleus RNA sequencing, the researchers combed through gene activity in thousands of individual brain cells. The technique revealed that Sst_Chodl neurons, a rare population of GABAergic cells that project long distances to other brain regions, showed the most severe molecular disruption. These cells are supposed to act as inhibitory brakes, calming down overactive circuits. Instead, they were firing too much.
Calcium imaging and patch-clamp recordings confirmed the finding. While many deep-layer somatostatin neurons showed blunted responses, Sst_Chodl cells bucked the trend, becoming hyperactive. The researchers then used a technique called chemogenetics to selectively silence these cells. When they did, the mice’s sleep normalized.
“This means that this type of brain cell plays a critical role in sleep in mice with this syndrome,” says Navneet Vasistha, an assistant professor at the university and one of the study’s lead authors. “Using a technique called chemogenetics, we can reduce the activity of these cells and restore normal sleep patterns, potentially alleviating other psychiatric symptoms as well.”
The finding is striking because Sst_Chodl neurons are so rare. They’re easy to miss in studies that lump all inhibitory cells together, yet they appear to be key regulators of circuit stability. Their long-range projections mean they can influence distant brain regions, making them potent mediators of widespread dysfunction.
Translating these results to humans will take years, if it’s possible at all. Mice aren’t people, and chemogenetics isn’t ready for clinical use. But the study offers a proof of principle: targeting a specific cell type can reverse a complex behavioral symptom. That’s a far cry from the blunt instruments currently available, which often come with side effects because they affect many brain regions at once.
“This cell type could potentially become a treatment target,” Vasistha says. “We hope that in the future, patients will benefit from a therapy for cognitive disorders that doesn’t broadly affect brain cells, but is so precisely targeted that side effects can be minimized.”
The research, published in Neuron, also underscores how much remains unknown about the cellular underpinnings of psychiatric disease. Schizophrenia is often framed as a disorder of dopamine or glutamate, the brain’s main excitatory neurotransmitter. But this study suggests that inhibitory neurons, particularly rare subtypes, may be just as important. And it hints that the critical period for intervention might come earlier than anyone thought, when the brain still has the plasticity to adapt.
“For a long time, the brain is able to compensate for developmental errors and maintain relatively normal function. But at some point, it’s like a chain snapping: the brain can no longer compensate, and that’s when symptoms emerge.”
“This cell type could potentially become a treatment target. We hope that in the future, patients will benefit from a therapy for cognitive disorders that doesn’t broadly affect brain cells, but is so precisely targeted that side effects can be minimized.”
Neuron: 10.1016/j.neuron.2025.08.028
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.