The white powder sits in a vacuum chamber, heating gently to 70°C. Nothing dramatic happens. No bubbling, no melting, no visible change at all. But peer closer at the molecular level and you’d witness something pharmaceutical companies have chased for decades: drug molecules transforming from rigid crystals into a disordered, highly soluble form, all without a drop of organic solvent.
It’s a surprisingly simple solution to what’s become known as the solubility crisis. Roughly 90 percent of drugs currently in development can’t dissolve well enough in water. For a pill to work, its active ingredients must first dissolve in your gastrointestinal fluids before entering your bloodstream. If a drug dissolves too slowly or not at all, it’s useless—no matter how brilliantly it works in theory. The pharmaceutical industry has watched billions in R&D investment dead-end here, promising compounds abandoned because they can’t make it past your stomach lining.
The conventional fix involves dissolving drugs in potent organic solvents like ethanol, then mixing them with porous materials that prevent recrystallisation. It works. But those solvents must be completely removed before the drug reaches patients, requiring expensive processing and raising environmental concerns. Residual solvents in formulations are strictly regulated. Some companies have spent years tweaking formulations just to eliminate the last traces.
Takehisa Hanawa at Tokyo University of Science wondered if there might be another way. His team focused on a physical quirk: some drugs sublime, transitioning directly from solid to gas without passing through a liquid phase. Could you use that vapour to load drugs onto porous carriers, skipping the solvent step entirely?
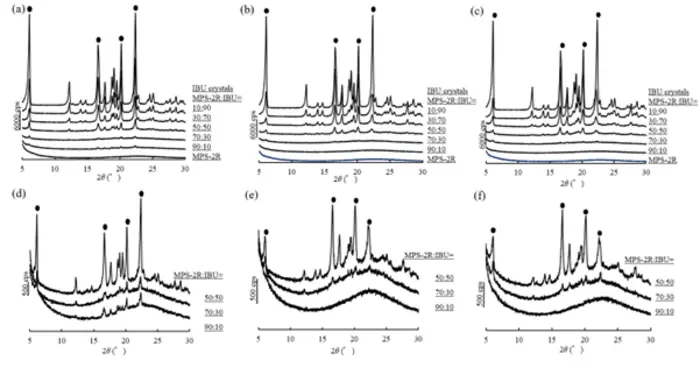
The researchers placed mesoporous silica—a cage-like material riddled with nanoscale pores—in a sealed container with ibuprofen powder. Under gentle heating and vacuum, the ibuprofen sublimed into gas, drifted through the chamber, and condensed into the silica’s pores, remaining amorphous rather than reforming crystals. No chemistry degree required to appreciate the elegance: solids become gas becomes trapped molecules.
When the team compared this sealed heating method with the conventional evaporation approach, the results were remarkably similar. Both techniques completely amorphised ibuprofen at low to moderate drug loadings, particularly when using silica with larger pore volumes. The crucial test came in dissolution experiments. Ibuprofen formulations prepared through sealed heating released the drug 2.7 times faster than raw crystalline ibuprofen within the first 10 minutes—matching the performance of solvent-made versions.
Chemical analyses confirmed the ibuprofen stayed intact throughout. The molecules didn’t react with the silica carrier or decompose during heating. This might seem obvious, but it’s not. Heat plus pharmaceuticals can produce unwanted reactions. That nothing broke down matters.
The sealed heating approach does have limits. It only works for drugs that sublime under reasonable conditions. Plenty of molecules don’t. But Hanawa sees broader potential. “By further adsorbing other pharmaceuticals onto MPS carrier-based compounds, we believe the proposed method could also be used to manufacture combination drugs,” he says. Load one drug through sealed heating, then potentially add another.
The environmental angle is equally compelling. “The fact that pharmaceuticals can be loaded onto MPS directly via the gas phase makes the SH method an environmentally friendly and safe drug loading technique that does not require the use of organic solvents,” Hanawa notes. No solvents means no solvent disposal, no purification steps to remove them, no regulatory hurdles around residual traces. Manufacturing becomes simpler.
The technique might work for other poorly soluble drugs beyond ibuprofen—candidates like aspirin, mefenamic acid, and various para-hydroxybenzoic acids. Each sublimes under the right conditions. Companies working with porous materials or mesoporous silica manufacturing could adopt the method relatively easily. The equipment isn’t exotic: vacuum chambers and heating elements, standard laboratory gear.
What’s striking about the research is how it reframes an old problem. Rather than fighting a drug’s tendency to crystallise by keeping it dissolved in harsh chemicals, you work with its physical properties—let it vaporise, let it condense where you want it. Sometimes the solution isn’t forcing molecules to behave differently, but finding a path that matches how they naturally move.
The pharmaceutical industry won’t transform overnight. Too many drugs don’t sublime, too many formulations are already optimised through conventional methods. But for the right compounds, particularly new ones still in development, this approach could remove a costly, wasteful step from manufacturing. And in an industry where 90 percent of pipeline drugs face solubility problems, any new tool matters.
Study link: https://jpharmsci.org/article/S0022-3549(25)00594-5/fulltext
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.